Coronaviruses
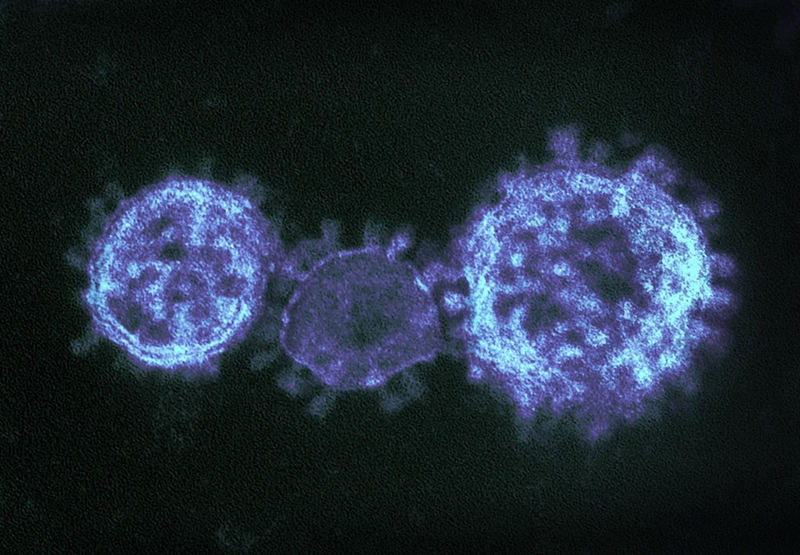
Coronaviruses are a large family of viruses that can infect a range of hosts. They are known to cause diseases including the common cold, Severe Acute Respiratory Syndrome (SARS) and Middle East Respiratory Syndrome (MERS) in humans.
In January 2020, China saw an outbreak of a new coronavirus strain now named SARS-CoV-2. Although the animal reservoir for the SARS and MERS viruses are known, this has yet to have been confirmed for SARS-CoV-2. All three strains are transmissible between humans.
To allow the widest possible distribution of relevant research, the Microbiology Society has brought together articles from across our portfolio and made this content freely available.
Image credit: "MERS-CoV" by NIAID is licensed under CC BY 2.0, this image has been modified.
Collection Contents
101 - 200 of 298 results
-
-
Discovery of a novel accessory protein NS7a encoded by porcine deltacoronavirus
More LessPorcine deltacoronavirus (PDCoV) is an emerging swine enteric coronavirus. Bioinformatics predicts that PDCoV encodes two accessory proteins (NS6 and NS7), the species-specific proteins for coronavirus. In this study, four mAbs against the predicted NS7 were prepared by using the purified recombinant NS7 protein. Indirect immunofluorescence assay demonstrated that all mAbs recognized cells transfected with an NS7 expression construct or infected with PDCoV. Western blot showed that NS7-specific mAbs recognized an additional protein band of about 12 kDa from PDCoV-infected cell lysates but not from cells with the ectopic expression of NS7. Detailed analysis suggested that this additional protein band represented a novel accessory protein, termed NS7a, a 100 amino acid polypeptide identical to the 3′ end of NS7. Moreover, NS7a is encoded by a separate subgenomic mRNA with a non-canonical transcription regulatory sequence. In summary, our results identified a third accessory protein encoded by PDCoV, which will enhance our understanding of PDCoV.
-
-
-
Feline coronavirus replication is affected by both cyclophilin A and cyclophilin B
More LessFeline coronavirus (FCoV) causes the fatal disease feline infectious peritonitis, which is currently incurable by drug treatment, and no effective vaccines are available. Cyclosporin A (CsA), a cyclophilin (Cyp) inhibitor, inhibits the replication of FCoV in vitro and in vivo as well as the replication of human and animal coronaviruses. However, the mechanism underlying the regulation of coronavirus replication by CsA is unknown. In this study, we analysed the role of Cyps in FCoV replication using knockdown and knockout cells specific to Cyps. Inhibition of CypA and CypB reduced FCoV replication, with replication in knockout cells being much less than that in knockdown cells. Furthermore, the proteins expressed by CypA and CypB harbouring mutations in their respective predicted peptidyl-prolyl cis–transisomerase active sites, which also alter the affinities between Cyps and CsA, inhibited FCoV replication. These findings indicate that the peptidyl-prolyl cis–transisomerase active sites of Cyps might be required for FCoV replication.
-
-
-
Naturally occurring recombination in ferret coronaviruses revealed by complete genome characterization
More LessFerret coronaviruses (FRCoVs) exist as an enteric and a systemic pathotype, of which the latter is highly lethal to ferrets. To our knowledge, this study provides the first full genome sequence of a FRCoV, tentatively called FRCoV-NL-2010, which was detected in 2010 in ferrets in The Netherlands. Phylogenetic analysis showed that FRCoV-NL-2010 is most closely related to mink CoV, forming a separate clade of mustelid alphacoronavirus that split off early from other alphacoronaviruses. Based on sequence homology of the complete genome, we propose that these mustelid coronaviruses may be assigned to a new species. Comparison of FRCoV-NL-2010 with the partially sequenced ferret systemic coronavirus MSU-1 and ferret enteric coronavirus MSU-2 revealed that recombination in the spike, 3c and envelope genes occurred between different FRCoVs.
-
-
-
The contribution of the cytoplasmic retrieval signal of severe acute respiratory syndrome coronavirus to intracellular accumulation of S proteins and incorporation of S protein into virus-like particles
More LessThe cytoplasmic tails of some coronavirus (CoV) spike (S) proteins contain an endoplasmic reticulum retrieval signal (ERRS) that can retrieve S proteins from the Golgi to the endoplasmic reticulum (ER); this process is thought to accumulate S proteins at the CoV budding site, the ER-Golgi intermediate compartment (ERGIC), and to facilitate S protein incorporation into virions. However, we showed previously that porcine epidemic diarrhoea CoV S proteins lacking the ERRS were efficiently incorporated into virions, similar to the original virus. Thus, the precise role of the ERRS in virus assembly remains unclear. Here, the roles of the S protein ERRS in severe acute respiratory syndrome CoV (SARS-CoV) intracellular trafficking and S incorporation into virus-like particles (VLPs) are described. Intracellular trafficking and indirect immunofluorescence analysis suggested that when M protein was present, wild-type S protein (wtS) could be retained in the pre- and post-medial Golgi compartments intracellularly and co-localized with M protein in the Golgi. In contrast, mutant S protein lacking the ERRS was distributed throughout the ER and only partially co-localized with M protein. Moreover, the intracellular accumulation of mutant S protein, particularly at the post-medial Golgi compartment, was significantly reduced compared with wtS. A VLP assay suggested that wtS that reached the post-medial compartment could be returned to the ERGIC for subsequent incorporation into VLPs, while mutant S protein could not. These results suggest that the ERRS of SARS-CoV contributes to intracellular S protein accumulation specifically in the post-medial Golgi compartment and to S protein incorporation into VLPs.
-
-
-
Evolution of infectious bronchitis virus in China over the past two decades
More LessAvian infectious bronchitis is a highly contagious disease caused by infectious bronchitis virus (IBV) that affects poultry production worldwide. The absence of vaccine cross-protection and the frequent emergence of new variant strains complicate control of IBV. Here we designed a study to measure the evolution dynamics of IBV strains in China. One hundered and seven complete sequences and 1022 S1-region sequences of Chinese IBVs isolated between 1994 and 2014 were analysed by using MEGA 5.0 software and the Bayesian analysis sampling trees (BEAST) method, and selection pressure on different proteins was assessed. The phylogenetic dissimilarity of different gene trees in the data set indicated possible recombination. Fourteen isolates were identified as recombinants, possibly generated from vaccines of the Massachusetts serotype in recombination with circulating viruses. The earliest IBV in China was found to have existed in the early 1900s, and continues to evolve at a rate of approximately 10− 5 substitutions per site per year. We found that purifying selection was the main evolutionary pressure in the protein-coding regions, while the S1 gene bears the greatest positive selection pressure. The proportion of QX-like genotype strains increased over time. These results indicate that the genotypes of Chinese IBVs have undergone a remarkable transition during the past 20 years.
-
-
-
Evidence for zoonotic origins of Middle East respiratory syndrome coronavirus
More LessHui-Ju Han, Hao Yu and Xue-Jie YuMiddle East respiratory syndrome (MERS) is an emerging infectious disease, caused by Middle East respiratory syndrome coronavirus (MERS-CoV) and is considered to be a zoonosis. However, the natural reservoirs of MERS-CoV remain obscure, with bats and camels as the most suspected sources. In this article, we review the evidence supporting a bat/camel origin of human MERS-CoV infection and current knowledge on the modes of camel-to-human transmission of MERS-CoV.
-
-
-
Middle East respiratory syndrome coronavirus shows poor replication but significant induction of antiviral responses in human monocyte-derived macrophages and dendritic cells
More LessIn this study we assessed the ability of Middle East respiratory syndrome coronavirus (MERS-CoV) to replicate and induce innate immunity in human monocyte-derived macrophages and dendritic cells (MDDCs), and compared it with severe acute respiratory syndrome coronavirus (SARS-CoV). Assessments of viral protein and RNA levels in infected cells showed that both viruses were impaired in their ability to replicate in these cells. Some induction of IFN-λ1, CXCL10 and MxA mRNAs in both macrophages and MDDCs was seen in response to MERS-CoV infection, but almost no such induction was observed in response to SARS-CoV infection. ELISA and Western blot assays showed clear production of CXCL10 and MxA in MERS-CoV-infected macrophages and MDDCs. Our data suggest that SARS-CoV and MERS-CoV replicate poorly in human macrophages and MDDCs, but MERS-CoV is nonetheless capable of inducing a readily detectable host innate immune response. Our results highlight a clear difference between the viruses in activating host innate immune responses in macrophages and MDDCs, which may contribute to the pathogenesis of infection.
-
-
-
First complete genome sequence of European turkey coronavirus suggests complex recombination history related with US turkey and guinea fowl coronaviruses
More LessA full-length genome sequence of 27 739 nt was determined for the only known European turkey coronavirus (TCoV) isolate. In general, the order, number and size of ORFs were consistent with other gammacoronaviruses. Three points of recombination were predicted, one towards the end of 1a, a second in 1b just upstream of S and a third in 3b. Phylogenetic analysis of the four regions defined by these three points supported the previous notion that European and American viruses do indeed have different evolutionary pathways. Very close relationships were revealed between the European TCoV and the European guinea fowl coronavirus in all regions except one, and both were shown to be closely related to the European infectious bronchitis virus (IBV) Italy 2005. None of these regions of sequence grouped European and American TCoVs. The region of sequence containing the S gene was unique in grouping all turkey and guinea fowl coronaviruses together, separating them from IBVs. Interestingly the French guinea fowl virus was more closely related to the North American viruses. These data demonstrate that European turkey and guinea fowl coronaviruses share a common genetic backbone (most likely an ancestor of IBV Italy 2005) and suggest that this recombined in two separate events with different, yet related, unknown avian coronaviruses, acquiring their S-3a genes. The data also showed that the North American viruses do not share a common backbone with European turkey and guinea fowl viruses; however, they do share similar S-3a genes with guinea fowl virus.
-
-
-
Isolation and identification of bat viruses closely related to human, porcine and mink orthoreoviruses
More LessBats have been identified as natural reservoirs of many viruses, including reoviruses. Recent studies have demonstrated the interspecies transmission of bat reoviruses to humans. In this study, we report the isolation and molecular characterization of six strains of mammalian orthoreovirus (MRV) from Hipposideros and Myotis spp. These isolates were grouped into MRV serotype 1, 2 or 3 based on the sequences of the S1 gene, which encodes the outer coat protein σ1. Importantly, we found that three of six bat MRV strains shared high similarity with MRVs isolated from diseased minks, piglets or humans based on the S1 segment, suggesting that interspecies transmission has occurred between bats and humans or animals. Phylogenetic analyses based on the 10 segments showed that the genomic segments of these bat MRVs had different evolution lineages, suggesting that these bat MRVs may have arisen through reassortment of MRVs of different origins.
-
-
-
The proteome of the infectious bronchitis virus Beau-R virion
More LessInfectious bronchitis is a highly contagious respiratory disease of poultry caused by the coronavirus infectious bronchitis virus (IBV). It was thought that coronavirus virions were composed of three major viral structural proteins until investigations of other coronaviruses showed that the virions also include viral non-structural and genus-specific accessory proteins as well as host-cell proteins. To study the proteome of IBV virions, virus was grown in embryonated chicken eggs, purified by sucrose-gradient ultracentrifugation and analysed by mass spectrometry. Analysis of three preparations of purified IBV yielded the three expected structural proteins plus 35 additional virion-associated host proteins. The virion-associated host proteins had a diverse range of functional attributions, being involved in cytoskeleton formation, RNA binding and protein folding pathways. Some of these proteins were unique to this study, while others were found to be orthologous to proteins identified in severe acute respiratory syndrome coronavirus virions and also virions from a number of other RNA and DNA viruses.
-
-
-
Arterivirus nsp12 versus the coronavirus nsp16 2′-O-methyltransferase: comparison of the C-terminal cleavage products of two nidovirus pp1ab polyproteins
More LessThe 3′-terminal domain of the most conserved ORF1b in three of the four families of the order Nidovirales (except for the family Arteriviridae) encodes a (putative) 2′-O-methyltransferase (2′-O-MTase), known as non structural protein (nsp) 16 in the family Coronaviridae and implicated in methylation of the 5′ cap structure of nidoviral mRNAs. As with coronavirus transcripts, arterivirus mRNAs are assumed to possess a 5′ cap although no candidate MTases have been identified thus far. To address this knowledge gap, we analysed the uncharacterized nsp12 of arteriviruses, which occupies the ORF1b position equivalent to that of the nidovirus 2′-O-MTase (coronavirus nsp16). In our in-depth bioinformatics analysis of nsp12, the protein was confirmed to be family specific whilst having diverged much further than other nidovirus ORF1b-encoded proteins, including those of the family Coronaviridae. Only one invariant and several partially conserved, predominantly aromatic residues were identified in nsp12, which may adopt a structure with alternating α-helices and β-strands, an organization also found in known MTases. However, no statistically significant similarity was found between nsp12 and the twofold larger coronavirus nsp16, nor could we detect MTase activity in biochemical assays using recombinant equine arteritis virus (EAV) nsp12. Our further analysis established that this subunit is essential for replication of this prototypic arterivirus. Using reverse genetics, we assessed the impact of 25 substitutions at 14 positions, yielding virus phenotypes ranging from WT-like to non-viable. Notably, replacement of the invariant phenylalanine 109 with tyrosine was lethal. We concluded that nsp12 plays an essential role during EAV replication, possibly by acting as a co-factor for another enzyme.
-
-
-
Genotyping coronaviruses associated with feline infectious peritonitis
More LessFeline coronavirus (FCoV) infections are endemic among cats worldwide. The majority of infections are asymptomatic or result in only mild enteric disease. However, approximately 5 % of cases develop feline infectious peritonitis (FIP), a systemic disease that is a frequent cause of death in young cats. In this study, we report the complete coding genome sequences of six FCoVs: three from faecal samples from healthy cats and three from tissue lesion samples from cats with confirmed FIP. The six samples were obtained over a period of 8 weeks at a single-site cat rescue and rehoming centre in the UK. We found amino acid differences located at 44 positions across an alignment of the six virus translatomes and, at 21 of these positions, the differences fully or partially discriminated between the genomes derived from the faecal samples and the genomes derived from the tissue lesion samples. In this study, two amino acid differences fully discriminated the two classes of genomes: these were both located in the S2 domain of the virus surface glycoprotein gene. We also identified deletions in the 3c protein ORF of genomes from two of the FIP samples. Our results support previous studies that implicate S protein mutations in the pathogenesis of FIP.
-
-
-
Phylodynamic analysis of avian infectious bronchitis virus in South America
More LessInfectious bronchitis virus (IBV) is a coronavirus of chickens that causes great economic losses to the global poultry industry. The present study focuses on South American IBVs and their genetic relationships with global strains. We obtained full-length sequences of the S1 coding region and N gene of IBV field isolates from Uruguay and Argentina, and performed Phylodynamic analysis to characterize the strains and estimate the time of the most recent common ancestor. We identified two major South American genotypes, which were here denoted South America I (SAI) and Asia/South America II (A/SAII). The SAI genotype is an exclusive South American lineage that emerged in the 1960s. The A/SAII genotype may have emerged in Asia in approximately 1995 before being introduced into South America. Both SAI and A/SAII genotype strains clearly differ from the Massachusetts strains that are included in the vaccine formulations being used in most South American countries.
-
-
-
A review of genetic methods and models for analysis of coronavirus-induced severe pneumonitis
More LessCoronaviruses (CoVs) have been studied for over 60 years, but have only recently gained notoriety as deadly human pathogens with the emergence of severe respiratory syndrome CoV and Middle East respiratory syndrome virus. The rapid emergence of these viruses has demonstrated the need for good models to study severe CoV respiratory infection and pathogenesis. There are, currently, different methods and models for the study of CoV disease. The available genetic methods for the study and evaluation of CoV genetics are reviewed here. There are several animal models, both mouse and alternative animals, for the study of severe CoV respiratory disease that have been examined, each with different pros and cons relative to the actual pathogenesis of the disease in humans. A current limitation of these models is that no animal model perfectly recapitulates the disease seen in humans. Through the review and analysis of the available disease models, investigators can employ the most appropriate available model to study various aspects of CoV pathogenesis and evaluate possible antiviral treatments that may potentially be successful in future treatment and prevention of severe CoV respiratory infections.
-
-
-
Role of the lipid rafts in the life cycle of canine coronavirus
More LessCoronaviruses are enveloped RNA viruses that have evolved complex relationships with their host cells, and modulate their lipid composition, lipid synthesis and signalling. Lipid rafts, enriched in sphingolipids, cholesterol and associated proteins, are special plasma membrane microdomains involved in several processes in viral infections. The extraction of cholesterol leads to disorganization of lipid microdomains and to dissociation of proteins bound to lipid rafts. Because cholesterol-rich microdomains appear to be a general feature of the entry mechanism of non-eneveloped viruses and of several coronaviruses, the purpose of this study was to analyse the contribution of lipids to the infectivity of canine coronavirus (CCoV). The CCoV life cycle is closely connected to plasma membrane cholesterol, from cell entry to viral particle production. The methyl-β-cyclodextrin (MβCD) was employed to remove cholesterol and to disrupt the lipid rafts. Cholesterol depletion from the cell membrane resulted in a dose-dependent reduction, but not abolishment, of virus infectivity, and at a concentration of 15 mM, the reduction in the infection rate was about 68 %. MβCD treatment was used to verify if cholesterol in the envelope was required for CCoV infection. This resulted in a dose-dependent inhibitory effect, and at a concentration of 9 mM MβCD, infectivity was reduced by about 73 %. Since viral entry would constitute a target for antiviral strategies, inhibitory molecules interacting with viral and/or cell membranes, or interfering with lipid metabolism, may have strong antiviral potential. It will be interesting in the future to analyse the membrane microdomains in the CCoV envelope.
-
-
-
Role of sialic acids in feline enteric coronavirus infections
More LessTo initiate infections, many coronaviruses use sialic acids, either as receptor determinants or as attachment factors helping the virus find its receptor underneath the heavily glycosylated mucus layer. In the present study, the role of sialic acids in serotype I feline enteric coronavirus (FECV) infections was studied in feline intestinal epithelial cell cultures. Treatment of cells with neuraminidase (NA) enhanced infection efficiency, showing that terminal sialic acid residues on the cell surface were not receptor determinants and even hampered efficient virus–receptor engagement. Knowing that NA treatment of coronaviruses can unmask viral sialic acid binding activity, replication of untreated and NA-treated viruses was compared, showing that NA treatment of the virus enhanced infectivity in untreated cells, but was detrimental in NA-treated cells. By using sialylated compounds as competitive inhibitors, it was demonstrated that sialyllactose (2,6-α-linked over 2,3-α-linked) notably reduced infectivity of NA-treated viruses, whereas bovine submaxillary mucin inhibited both treated and untreated viruses. In desialylated cells, however, viruses were less prone to competitive inhibition with sialylated compounds. In conclusion, this study demonstrated that FECV had a sialic acid binding capacity, which was partially masked by virus-associated sialic acids, and that attachment to sialylated compounds could facilitate enterocyte infections. However, sialic acid binding was not a prerequisite for the initiation of infection and virus–receptor engagement was even more efficient after desialylation of cells, indicating that FECV requires sialidases for efficient enterocyte infections.
-
-
-
The ORF4b-encoded accessory proteins of Middle East respiratory syndrome coronavirus and two related bat coronaviruses localize to the nucleus and inhibit innate immune signalling
More LessThe recently emerged Middle East respiratory syndrome coronavirus (MERS-CoV), a betacoronavirus, is associated with severe pneumonia and renal failure. The environmental origin of MERS-CoV is as yet unknown; however, its genome sequence is closely related to those of two bat coronaviruses, named BtCoV-HKU4 and BtCoV-HKU5, which were derived from Chinese bat samples. A hallmark of highly pathogenic respiratory viruses is their ability to evade the innate immune response of the host. CoV accessory proteins, for example those from severe acute respiratory syndrome CoV (SARS-CoV), have been shown to block innate antiviral signalling pathways. MERS-CoV, similar to SARS-CoV, has been shown to inhibit type I IFN induction in a variety of cell types in vitro. We therefore hypothesized that MERS-CoV and the phylogenetically related BtCoV-HKU4 and BtCoV-HKU5 may encode proteins with similar capabilities. In this study, we have demonstrated that the ORF4b-encoded accessory protein (p4b) of MERS-CoV, BtCoV-HKU4 and BtCoV-HKU5 may indeed facilitate innate immune evasion by inhibiting the type I IFN and NF-κB signalling pathways. We also analysed the subcellular localization of p4b from MERS-CoV, BtCoV-HKU4 and BtCoV-HKU5 and demonstrated that all are localized to the nucleus.
-
-
-
Interferon-β and mycophenolic acid are potent inhibitors of Middle East respiratory syndrome coronavirus in cell-based assays
More LessThe Middle East respiratory syndrome coronavirus (MERS-CoV) presents a novel emerging threat to public health worldwide. Several treatments for infected individuals have been suggested including IFN, ribavirin and passive immunotherapy with convalescent plasma. Administration of IFN-α2b and ribavirin has improved outcomes of MERS-CoV infection in rhesus macaques when administered within 8 h post-challenge. However, detailed and systematic evidence on the activity of other clinically available drugs is limited. Here we compared the susceptibility of MERS-CoV with different IFN products (IFN-α2b, IFN-γ, IFN-universal, IFN-α2a and IFN-β), as well as with two antivirals, ribavirin and mycophenolic acid (MPA), against MERS-CoV (Hu/Jordan-N3/2012) in vitro. Of all the IFNs tested, IFN-β showed the strongst inhibition of MERS-CoV in vitro, with an IC50 of 1.37 U ml−1, 41 times lower than the previously reported IC50 (56.08 U ml−1) of IFN-α2b. IFN-β inhibition was confirmed in the virus yield reduction assay, with an IC90 of 38.8 U ml−1. Ribavirin did not inhibit viral replication in vitro at a dose that would be applicable to current treatment protocols in humans. In contrast, MPA showed strong inhibition, with an IC50 of 2.87 µM. This drug has not been previously tested against MERS-CoV and may provide an alternative to ribavirin for treatment of MERS-CoV. In conclusion, IFN-β, MPA or a combination of the two may be beneficial in the treatment of MERS-CoV or as a post-exposure intervention in high-risk patients with known exposures to MERS-CoV.
-
-
-
Neutrophils are needed for an effective immune response against pulmonary rat coronavirus infection, but also contribute to pathology
More LessPolymorphonuclear neutrophils (PMN) infiltrate the respiratory tract early after viral infection and can contribute to both host defence and pathology. Coronaviruses are important causes of respiratory tract infections, ranging from mild to severe depending on the viral strain. This study evaluated the role of PMN during a non-fatal pulmonary coronavirus infection in the natural host. Rat coronavirus (RCoV) causes respiratory disease in adult rats, characterized by an early PMN response, viral replication and inflammatory lesions in the lungs, mild weight loss and effective resolution of infection. To determine their role during RCoV infection, PMN were depleted and the effects on disease progression, viral replication, inflammatory response and lung pathology were analysed. Compared with RCoV infection in control animals, PMN-depleted rats had worsened disease with weight loss, clinical signs, mortality and prolonged pulmonary viral replication. PMN-depleted animals had fewer macrophages and lymphocytes in the respiratory tract, corresponding to lower chemokine levels. Combined with in vitro experiments showing that PMN express cytokines and chemokines in response to RCoV-infected alveolar epithelial cells, these findings support a role for PMN in eliciting an inflammatory response to RCoV infection. Despite their critical role in the protection from severe disease, the presence of PMN was correlated with haemorrhagic lesions, epithelial barrier permeability and cellular inflammation in the lungs. This study demonstrated that while PMN are required for an effective antiviral response, they also contribute to lung pathology during RCoV infection.
-
-
-
Proteolytic processing, deubiquitinase and interferon antagonist activities of Middle East respiratory syndrome coronavirus papain-like protease
More LessThe emerging Middle East respiratory syndrome coronavirus (MERS-CoV) causes severe pulmonary disease in humans and represents the second example of a highly pathogenic coronavirus (CoV) following severe acute respiratory syndrome coronavirus (SARS-CoV). Genomic studies revealed that two viral proteases, papain-like protease (PLpro) and 3C-like protease (3CLpro), process the polyproteins encoded by the MERS-CoV genomic RNA. We previously reported that SARS-CoV PLpro acts as both deubiquitinase (DUB) and IFN antagonist, but the function of the MERS-CoV PLpro was poorly understood. In this study, we characterized MERS-CoV PLpro, which is a protease and can recognize and process the cleavage sites (CS) of nsp1-2, nsp2-3 and nsp3-4. The LXGG consensus cleavage sites in the N terminus of pp1a/1ab, which is generally essential for CoV PLpro-mediated processing, were also characterized in MERS-CoV. MERS-CoV PLpro, like human SARS-CoV PLpro and NL63-CoV PLP2, is a viral deubiquitinating enzyme. It acts on both K48- and K63-linked ubiquitination and ISG15-linked ISGylation. We confirmed that MERS-CoV PLpro acts as an IFN antagonist through blocking the phosphorylation and nuclear translocation of IFN regulatory factor 3 (IRF3). These findings indicate that MERS-CoV PLpro acts as a viral DUB and suppresses production of IFN-β by an interfering IRF3-mediated signalling pathway, in addition to recognizing and processing the CS at the N terminus of replicase polyprotein to release the non-structural proteins. The characterization of proteolytic processing, DUB and IFN antagonist activities of MERS-CoV PLpro would reveal the interactions between MERS-CoV and its host, and be applicable to develop strategies targeting PLpro for the effective control of MERS-CoV infection.
-
-
-
Wild-type and innate immune-deficient mice are not susceptible to the Middle East respiratory syndrome coronavirus
More LessThe Middle East respiratory syndrome coronavirus (MERS-CoV) is a newly emerging highly pathogenic virus causing almost 50 % lethality in infected individuals. The development of a small-animal model is critical for the understanding of this virus and to aid in development of countermeasures against MERS-CoV. We found that BALB/c, 129/SvEv and 129/SvEv STAT1 knockout mice are not permissive to MERS-CoV infection. The lack of infection may be due to the low level of mRNA and protein for the MERS-CoV receptor, dipeptidyl peptidase 4 (DPP4), in the lungs of mice. The low level of DPP4 in the lungs likely contributes to the lack of viral replication in these mouse models and suggests that a transgenic mouse model expressing DPP4 to higher levels is necessary to create a mouse model for MERS-CoV.
-
-
-
Highly diversified coronaviruses in neotropical bats
More LessVictor Max Corman, Andrea Rasche, Thierno Diawo Diallo, Veronika M. Cottontail, Andreas Stöcker, Breno Frederico de Carvalho Dominguez Souza, Jefferson Ivan Corrêa, Aroldo José Borges Carneiro, Carlos Roberto Franke, Martina Nagy, Markus Metz, Mirjam Knörnschild, Elisabeth K. V. Kalko, Simon J. Ghanem, Karen D. Sibaja Morales, Egoitz Salsamendi, Manuel Spínola, Georg Herrler, Christian C. Voigt, Marco Tschapka, Christian Drosten and Jan Felix DrexlerBats host a broad diversity of coronaviruses (CoVs), including close relatives of human pathogens. There is only limited data on neotropical bat CoVs. We analysed faecal, blood and intestine specimens from 1562 bats sampled in Costa Rica, Panama, Ecuador and Brazil for CoVs by broad-range PCR. CoV RNA was detected in 50 bats representing nine different species, both frugivorous and insectivorous. These bat CoVs were unrelated to known human or animal pathogens, indicating an absence of recent zoonotic spill-over events. Based on RNA-dependent RNA polymerase (RdRp)-based grouping units (RGUs) as a surrogate for CoV species identification, the 50 viruses represented five different alphacoronavirus RGUs and two betacoronavirus RGUs. Closely related alphacoronaviruses were detected in Carollia perspicillata and C. brevicauda across a geographical distance exceeding 5600 km. Our study expands the knowledge on CoV diversity in neotropical bats and emphasizes the association of distinct CoVs and bat host genera.
-
-
-
MERS-coronavirus replication induces severe in vitro cytopathology and is strongly inhibited by cyclosporin A or interferon-α treatment
More LessCoronavirus (CoV) infections are commonly associated with respiratory and enteric disease in humans and animals. The 2003 outbreak of severe acute respiratory syndrome (SARS) highlighted the potentially lethal consequences of CoV-induced disease in humans. In 2012, a novel CoV (Middle East Respiratory Syndrome coronavirus; MERS-CoV) emerged, causing 49 human cases thus far, of which 23 had a fatal outcome. In this study, we characterized MERS-CoV replication and cytotoxicity in human and monkey cell lines. Electron microscopy of infected Vero cells revealed extensive membrane rearrangements, including the formation of double-membrane vesicles and convoluted membranes, which have been implicated previously in the RNA synthesis of SARS-CoV and other CoVs. Following infection, we observed rapidly increasing viral RNA synthesis and release of high titres of infectious progeny, followed by a pronounced cytopathology. These characteristics were used to develop an assay for antiviral compound screening in 96-well format, which was used to identify cyclosporin A as an inhibitor of MERS-CoV replication in cell culture. Furthermore, MERS-CoV was found to be 50–100 times more sensitive to alpha interferon (IFN-α) treatment than SARS-CoV, an observation that may have important implications for the treatment of MERS-CoV-infected patients. MERS-CoV infection did not prevent the IFN-induced nuclear translocation of phosphorylated STAT1, in contrast to infection with SARS-CoV where this block inhibits the expression of antiviral genes. These findings highlight relevant differences between these distantly related zoonotic CoVs in terms of their interaction with and evasion of the cellular innate immune response.
-
-
-
Coronaviruses in bats from Mexico
More LessBats are reservoirs for a wide range of human pathogens including Nipah, Hendra, rabies, Ebola, Marburg and severe acute respiratory syndrome coronavirus (CoV). The recent implication of a novel beta (β)-CoV as the cause of fatal respiratory disease in the Middle East emphasizes the importance of surveillance for CoVs that have potential to move from bats into the human population. In a screen of 606 bats from 42 different species in Campeche, Chiapas and Mexico City we identified 13 distinct CoVs. Nine were alpha (α)-CoVs; four were β-CoVs. Twelve were novel. Analyses of these viruses in the context of their hosts and ecological habitat indicated that host species is a strong selective driver in CoV evolution, even in allopatric populations separated by significant geographical distance; and that a single species/genus of bat can contain multiple CoVs. A β-CoV with 96.5 % amino acid identity to the β-CoV associated with human disease in the Middle East was found in a Nyctinomops laticaudatus bat, suggesting that efforts to identify the viral reservoir should include surveillance of the bat families Molossidae/Vespertilionidae, or the closely related Nycteridae/Emballonuridae. While it is important to investigate unknown viral diversity in bats, it is also important to remember that the majority of viruses they carry will not pose any clinical risk, and bats should not be stigmatized ubiquitously as significant threats to public health.
-
-
-
Combined action of type I and type III interferon restricts initial replication of severe acute respiratory syndrome coronavirus in the lung but fails to inhibit systemic virus spread
More LessSTAT1-deficient mice are more susceptible to infection with severe acute respiratory syndrome coronavirus (SARS-CoV) than type I interferon (IFN) receptor-deficient mice. We used mice lacking functional receptors for both type I and type III IFN (double knockout, dKO) to evaluate the possibility that type III IFN plays a decisive role in SARS-CoV protection. We found that viral peak titres in lungs of dKO and STAT1-deficient mice were similar, but significantly higher than in wild-type mice. The kinetics of viral clearance from the lung were also comparable in dKO and STAT1-deficient mice. Surprisingly, however, infected dKO mice remained healthy, whereas infected STAT1-deficient mice developed liver pathology and eventually succumbed to neurological disease. Our data suggest that the failure of STAT1-deficient mice to control initial SARS-CoV replication efficiently in the lung is due to impaired type I and type III IFN signalling, whereas the failure to control subsequent systemic viral spread is due to unrelated defects in STAT1-deficient mice.
-
-
-
Genomic analysis of 16 Colorado human NL63 coronaviruses identifies a new genotype, high sequence diversity in the N-terminal domain of the spike gene and evidence of recombination
More LessThis study compared the complete genome sequences of 16 NL63 strain human coronaviruses (hCoVs) from respiratory specimens of paediatric patients with respiratory disease in Colorado, USA, and characterized the epidemiology and clinical characteristics associated with circulating NL63 viruses over a 3-year period. From 1 January 2009 to 31 December 2011, 92 of 9380 respiratory specimens were found to be positive for NL63 RNA by PCR, an overall prevalence of 1 %. NL63 viruses were circulating during all 3 years, but there was considerable yearly variation in prevalence and the month of peak incidence. Phylogenetic analysis comparing the genome sequences of the 16 Colorado NL63 viruses with those of the prototypical hCoV-NL63 and three other NL63 viruses from the Netherlands demonstrated that there were three genotypes (A, B and C) circulating in Colorado from 2005 to 2010, and evidence of recombination between virus strains was found. Genotypes B and C co-circulated in Colorado in 2005, 2009 and 2010, but genotype A circulated only in 2005 when it was the predominant NL63 strain. Genotype C represents a new lineage that has not been described previously. The greatest variability in the NL63 virus genomes was found in the N-terminal domain (NTD) of the spike gene (nt 1–600, aa 1–200). Ten different amino acid sequences were found in the NTD of the spike protein among these NL63 strains and the 75 partial published sequences of NTDs from strains found at different times throughout the world.
-
-
-
Organ tropism of murine coronavirus does not correlate with the expression levels of the membrane-anchored or secreted isoforms of the carcinoembryonic antigen-related cell adhesion molecule 1 receptor
More LessCarcinoembryonic antigen-related cell adhesion molecule 1 (CEACAM1) is the sole known receptor of murine hepatitis virus (MHV) A59, but the available, often qualitative, data about CEACAM1 expression does not explain MHV organ tropism. Ceacam1 transcripts undergo alternative splicing resulting in multiple isoforms, including secreted CEACAM1 isoforms that can neutralize the virus. We determined the quantities of Ceacam1 transcripts encoding membrane-bound and secreted isoforms in mouse organs and a set of cell lines. In vivo, the lowest receptor mRNA levels were found in brain and muscle and these were similar to those in easily infectable cultured cells. While the quantities of the receptor transcripts varied between mouse organs, their abundance did not correlate with susceptibility to MHV infection. The proportion of transcripts encoding secreted isoforms also could not explain the selection of sites for virus replication, as it was constant in all organs. Our data suggest that neither of the two CEACAM1 isoforms defines MHV organ tropism.
-
-
-
Replication-dependent downregulation of cellular angiotensin-converting enzyme 2 protein expression by human coronavirus NL63
More LessLike severe acute respiratory syndrome coronavirus (SARS-CoV), human coronavirus (HCoV)-NL63 employs angiotensin-converting enzyme 2 (ACE2) as a receptor for cellular entry. SARS-CoV infection causes robust downregulation of cellular ACE2 expression levels and it has been suggested that the SARS-CoV effect on ACE2 is involved in the severity of disease. We investigated whether cellular ACE2 downregulation occurs at optimal replication conditions of HCoV-NL63 infection. The expression of the homologue of ACE2, the ACE protein not used as a receptor by HCoV-NL63, was measured as a control. A specific decrease for ACE2 protein level was observed when HCoV-NL63 was cultured at 34 °C. Culturing the virus at the suboptimal temperature of 37 °C resulted in low replication of the virus and the effect on ACE2 expression was lost. We conclude that the decline of ACE2 expression is dependent on the efficiency of HCoV-NL63 replication, and that HCoV-NL63 and SARS-CoV both affect cellular ACE2 expression during infection.
-
-
-
Two palmitylated cysteine residues of the severe acute respiratory syndrome coronavirus spike (S) protein are critical for S incorporation into virus-like particles, but not for M–S co-localization
More LessThe endodomain of several coronavirus (CoV) spike (S) proteins contains palmitylated cysteine residues and enables co-localization and interaction with the CoV membrane (M) protein. Depalmitylation of mouse hepatitis virus S proteins abolished this interaction, resulting in the failure of S incorporation into virions. In contrast, an immunofluorescence assay (IFA) showed that depalmitylated severe acute respiratory syndrome coronavirus (SCoV) S proteins still co-localized with the M protein in the budding site. Here, we determined the ability of depalmitylated SCoV S mutants to incorporate S into virus-like particles (VLPs). IFA confirmed that all SCoV S mutants co-localized with the M protein intracellularly. However, the mutants lacking two cysteine residues (C1234/1235) failed to incorporate S into VLPs. This indicated that these palmitylated cysteines are essential for S incorporation, but are not involved in S co-localization mediated by the M protein. Our findings suggest that M–S co-localization and S incorporation occur independently of one another in SCoV virion assembly.
-
-
-
Infection of human alveolar macrophages by human coronavirus strain 229E
More LessHuman coronavirus strain 229E (HCoV-229E) commonly causes upper respiratory tract infections. However, lower respiratory tract infections can occur in some individuals, indicating that cells in the distal lung are susceptible to HCoV-229E. This study determined the virus susceptibility of primary cultures of human alveolar epithelial cells and alveolar macrophages (AMs). Fluorescent antibody staining indicated that HCoV-229E could readily infect AMs, but no evidence was found for infection in differentiated alveolar epithelial type II cells and only a very low level of infection in type II cells transitioning to the type I-like cell phenotype. However, a human bronchial epithelial cell line (16HBE) was readily infected. The innate immune response of AMs to HCoV-229E infection was evaluated for cytokine production and interferon (IFN) gene expression. AMs secreted significant amounts of tumour necrosis factor alpha (TNF-α), regulated on activation normal T-cell expressed and secreted (RANTES/CCL5) and macrophage inflammatory protein 1β (MIP-1β/CCL4) in response to HCoV-229E infection, but these cells exhibited no detectable increase in IFN-β or interleukin-29 in mRNA levels. AMs from smokers had reduced secretion of TNF-α compared with non-smokers in response to HCoV-229E infection. Surfactant protein A (SP-A) and SP-D are part of the innate immune system in the distal lung. Both surfactant proteins bound to HCoV-229E, and pre-treatment of HCoV-229E with SP-A or SP-D inhibited infection of 16HBE cells. In contrast, there was a modest reduction in infection in AMs by SP-A, but not by SP-D. In summary, AMs are an important target for HCoV-229E, and they can mount a pro-inflammatory innate immune response to infection.
-
-
-
Bat severe acute respiratory syndrome-like coronavirus ORF3b homologues display different interferon antagonist activities
More LessThe ORF3b protein of severe acute respiratory syndrome coronavirus (SARS-CoV) has a nuclear localization signal (NLS) at its C terminus and antagonizes interferon (IFN) function by modulating the activity of IFN regulatory factor 3 (IRF3). SARS-like coronaviruses (SL-CoVs) found in bats share an identical genome organization and high sequence identity for most of their gene products. In this study, ORF3b homologues were identified from three bat SL-CoV strains. These ORF3b homologues were C-terminally truncated and lacked the C-terminal NLS of SARS-CoV. IFN antagonist activities analysis demonstrated that one SL-CoV ORF3b still possessed IFN antagonist and IRF3-modulating activities. These results indicate that different ORF3b proteins display different IFN antagonist activities and this function is independent of the protein’s nuclear localization, suggesting a potential link between bat SL-CoV ORF3b function and viral pathogenesis.
-
-
-
Cyclosporin A inhibits the replication of diverse coronaviruses
More LessLow micromolar, non-cytotoxic concentrations of cyclosporin A (CsA) strongly affected the replication of severe acute respiratory syndrome coronavirus (SARS-CoV), human coronavirus 229E and mouse hepatitis virus in cell culture, as was evident from the strong inhibition of GFP reporter gene expression and a reduction of up to 4 logs in progeny titres. Upon high-multiplicity infection, CsA treatment rendered SARS-CoV RNA and protein synthesis almost undetectable, suggesting an early block in replication. siRNA-mediated knockdown of the expression of the prominent CsA targets cyclophilin A and B did not affect SARS-CoV replication, suggesting either that these specific cyclophilin family members are dispensable or that the reduced expression levels suffice to support replication.
-
-
-
The ADP-ribose-1″-monophosphatase domains of severe acute respiratory syndrome coronavirus and human coronavirus 229E mediate resistance to antiviral interferon responses
More LessSeveral plus-strand RNA viruses encode proteins containing macrodomains. These domains possess ADP-ribose-1″-phosphatase (ADRP) activity and/or bind poly(ADP-ribose), poly(A) or poly(G). The relevance of these activities in the viral life cycle has not yet been resolved. Here, we report that genetically engineered mutants of severe acute respiratory syndrome coronavirus (SARS-CoV) and human coronavirus 229E (HCoV-229E) expressing ADRP-deficient macrodomains displayed an increased sensitivity to the antiviral effect of alpha interferon compared with their wild-type counterparts. The data suggest that macrodomain-associated ADRP activities may have a role in viral escape from the innate immune responses of the host.
-
-
-
Infection with human coronavirus NL63 enhances streptococcal adherence to epithelial cells
More LessUnderstanding the mechanisms of augmented bacterial pathogenicity in post-viral infections is the first step in the development of an effective therapy. This study assessed the effect of human coronavirus NL63 (HCoV-NL63) on the adherence of bacterial pathogens associated with respiratory tract illnesses. It was shown that HCoV-NL63 infection resulted in an increased adherence of Streptococcus pneumoniae to virus-infected cell lines and fully differentiated primary human airway epithelium cultures. The enhanced binding of bacteria correlated with an increased expression level of the platelet-activating factor receptor (PAF-R), but detailed evaluation of the bacterium–PAF-R interaction revealed a limited relevance of this process.
-
-
-
Severe acute respiratory syndrome coronavirus papain-like protease suppressed alpha interferon-induced responses through downregulation of extracellular signal-regulated kinase 1-mediated signalling pathways
More LessSevere acute respiratory syndrome coronavirus (SARS-CoV) papain-like protease (PLpro), a deubiquitinating enzyme, reportedly blocks poly I : C-induced activation of interferon regulatory factor 3 and nuclear factor kappa B, reducing interferon (IFN) induction. This study investigated type I IFN antagonist mechanism of PLpro in human promonocytes. PLpro antagonized IFN-α-induced responses such as interferon-stimulated response element- and AP-1-driven promoter activation, protein kinase R, 2′-5′-oligoadenylate synthetase (OAS), interleukin (IL)-6 and IL-8 expression, and signal transducers and activators of transcription (STAT) 1 (Tyr701), STAT1 (Ser727) and c-Jun phosphorylation. A proteomics approach demonstrated downregulation of extracellular signal-regulated kinase (ERK) 1 and upregulation of ubiquitin-conjugating enzyme (UBC) E2-25k as inhibitory mechanism of PLpro on IFN-α-induced responses. IFN-α treatment significantly induced mRNA expression of UBC E2-25k, but not ERK1, causing time-dependent decrease of ERK1, but not ERK2, in PLpro-expressing cells. Poly-ubiquitination of ERK1 showed a relationship between ERK1 and ubiquitin proteasome signalling pathways associated with IFN antagonism by PLpro. Combination treatment of IFN-α and the proteasome inhibitor MG-132 showed a time-dependent restoration of ERK1 protein levels and significant increase of ERK1, STAT1 and c-Jun phosphorylation in PLpro-expressing cells. Importantly, PD098059 (an ERK1/2 inhibitor) treatment significantly reduced IFN-α-induced ERK1 and STAT1 phosphorylation, inhibiting IFN-α-induced expression of 2′-5′-OAS in vector control cells and PLpro-expressing cells. Overall results proved downregulation of ERK1 by ubiquitin proteasomes and suppression of interaction between ERK1 and STAT1 as type I IFN antagonist function of SARS-CoV PLpro.
-
-
-
Identification of key amino acid residues required for horseshoe bat angiotensin-I converting enzyme 2 to function as a receptor for severe acute respiratory syndrome coronavirus
More LessAngiotensin-I converting enzyme 2 (ACE2) is the receptor for severe acute respiratory syndrome (SARS) coronavirus (SARS-CoV). A previous study indicated that ACE2 from a horseshoe bat, the host of a highly related SARS-like coronavirus, could not function as a receptor for SARS-CoV. Here, we demonstrate that a 3 aa change from SHE (aa 40–42) to FYQ was sufficient to convert the bat ACE2 into a fully functional receptor for SARS-CoV. We further demonstrate that an ACE2 molecule from a fruit bat, which contains the FYQ motif, was able to support SARS-CoV infection, indicating a potentially much wider host range for SARS-CoV-related viruses among different bat populations.
-
-
-
Sites of feline coronavirus persistence in healthy cats
More LessFeline coronavirus (FCoV) is transmitted via the faecal–oral route and primarily infects enterocytes, but subsequently spreads by monocyte-associated viraemia. In some infected cats, virulent virus mutants induce feline infectious peritonitis (FIP), a fatal systemic disease that can develop in association with viraemia. Persistently infected, healthy carriers are believed to be important in the epidemiology of FIP, as they represent a constant source of FCoV, shed either persistently or intermittently in faeces. So far, the sites of virus persistence have not been determined definitely. The purpose of this study was to examine virus distribution and viral load in organs and gut compartments of specified-pathogen-free cats, orally infected with non-virulent type I FCoV, over different time periods and with or without detectable viraemia. The colon was identified as the major site of FCoV persistence and probable source for recurrent shedding, but the virus was shown also to persist in several other organs, mainly in tissue macrophages. These might represent additional sources for recurrent viraemia.
-
-
-
Intraspecies diversity of SARS-like coronaviruses in Rhinolophus sinicus and its implications for the origin of SARS coronaviruses in humans
More LessThe Chinese rufous horseshoe bat (Rhinolophus sinicus) has been suggested to carry the direct ancestor of severe acute respiratory syndrome (SARS) coronavirus (SCoV), and the diversity of SARS-like CoVs (SLCoV) within this Rhinolophus species is therefore worth investigating. Here, we demonstrate the remarkable diversity of SLCoVs in R. sinicus and identify a strain with the same pattern of phylogenetic incongruence (i.e. an indication of recombination) as reported previously in another SLCoV strain. Moreover, this strain possesses a distinctive 579 nt deletion in the nsp3 region that was also found in a human SCoV from the late-phase epidemic. Phylogenetic analysis of the Orf1 region suggested that the human SCoVs are phylogenetically closer to SLCoVs in R. sinicus than to SLCoVs in other Rhinolophus species. These findings reveal a closer evolutionary linkage between SCoV in humans and SLCoVs in R. sinicus, defining the scope of surveillance to search for the direct ancestor of human SCoVs.
-
-
-
Feline infectious peritonitis: insights into feline coronavirus pathobiogenesis and epidemiology based on genetic analysis of the viral 3c gene
More LessFeline infectious peritonitis (FIP) is a lethal systemic disease caused by FIP virus (FIPV), a virulent mutant of apathogenic feline enteric coronavirus (FECV). We analysed the 3c gene – a proposed virulence marker – in 27 FECV- and 28 FIPV-infected cats. Our findings suggest that functional 3c protein expression is crucial for FECV replication in the gut, but dispensable for systemic FIPV replication. Whilst intact in all FECVs, the 3c gene was mutated in the majority (71.4 %) of FIPVs, but not in all, implying that mutation in 3c is not the (single) cause of FIP. Most cats with FIP had no detectable intestinal feline coronaviruses (FCoVs) and had seemingly cleared the primary FECV infection. In those with detectable intestinal FCoV, the virus always had an intact 3c and seemed to have been acquired by FECV superinfection. Apparently, 3c-inactivated viruses replicate not at all – or only poorly – in the gut, explaining the rare incidence of FIP outbreaks.
-
-
-
Rat respiratory coronavirus infection: replication in airway and alveolar epithelial cells and the innate immune response
More LessThe rat coronavirus sialodacryoadenitis virus (SDAV) causes respiratory infection and provides a system for investigating respiratory coronaviruses in a natural host. A viral suspension in the form of a microspray aerosol was delivered by intratracheal instillation into the distal lung of 6–8-week-old Fischer 344 rats. SDAV inoculation produced a 7 % body weight loss over a 5 day period that was followed by recovery over the next 7 days. SDAV caused focal lesions in the lung, which were most severe on day 4 post-inoculation (p.i.). Immunofluorescent staining showed that four cell types supported SDAV virus replication in the lower respiratory tract, namely Clara cells, ciliated cells in the bronchial airway and alveolar type I and type II cells in the lung parenchyma. In bronchial alveolar lavage fluid (BALF) a neutrophil influx increased the population of neutrophils to 45 % compared with 6 % of the cells in control samples on day 2 after mock inoculation. Virus infection induced an increase in surfactant protein SP-D levels in BALF of infected rats on days 4 and 8 p.i. that subsided by day 12. The concentrations of chemokines MCP-1, LIX and CINC-1 in BALF increased on day 4 p.i., but returned to control levels by day 8. Intratracheal instillation of rats with SDAV coronavirus caused an acute, self-limited infection that is a useful model for studying the early events of the innate immune response to respiratory coronavirus infections in lungs of the natural virus host.
-
-
-
Angiotensin-converting enzyme 2 (ACE2) from raccoon dog can serve as an efficient receptor for the spike protein of severe acute respiratory syndrome coronavirus
More LessRaccoon dog is one of the suspected intermediate hosts of severe acute respiratory syndrome coronavirus (SARS-CoV). In this study, the angiotensin-converting enzyme 2 (ACE2) gene of raccoon dog (rdACE2) was cloned and sequenced. The amino acid sequence of rdACE2 has identities of 99.3, 89.2, 83.9 and 80.4 % to ACE2 proteins from dog, masked palm civet (pcACE2), human (huACE2) and bat, respectively. There are six amino acid changes in rdACE2 compared with huACE2, and four changes compared with pcACE2, within the 18 residues of ACE2 known to make direct contact with the SARS-CoV S protein. A HeLa cell line stably expressing rdACE2 was established; Western blot analyses and an enzyme-activity assay indicated that the cell line expressed ACE2 at a similar level to two previously established cell lines that express ACE2 from human and masked palm civet, respectively. Human immunodeficiency virus-backboned pseudoviruses expressing spike proteins derived from human SARS-CoV or SARS-CoV-like viruses of masked palm civets and raccoon dogs were tested for their entry efficiency into these cell lines. The results showed that rdACE2 is a more efficient receptor for human SARS-CoV, but not for SARS-CoV-like viruses of masked palm civets and raccoon dogs, than huACE2 or pcACE2. This study provides useful data to elucidate the role of raccoon dog in SARS outbreaks.
-
-
-
Severe acute respiratory syndrome coronavirus nucleocapsid protein does not modulate transcription of the human FGL2 gene
More LessAmong the structural and nonstructural proteins of severe acute respiratory syndrome coronavirus (SARS-CoV), the nucleocapsid (N) protein plays pivotal roles in the biology and pathogenesis of viral infection. N protein is thought to dysregulate cell signalling and the transcription of cellular genes, including FGL2, which encodes a prothrombinase implicated in vascular thrombosis, fibrin deposition and pneumocyte necrosis. Here, we showed that N protein expressed in cultured human cells was predominantly found in the cytoplasm and was competent in repressing the transcriptional activity driven by interferon-stimulated response elements. However, the expression of N protein did not influence the transcription from the FGL2 promoter. More importantly, N protein did not modulate the expression of FGL2 mRNA or protein in transfected or SARS-CoV-infected cells. Taken together, our findings did not support the model in which SARS-CoV N protein specifically modulates transcription of the FGL2 gene to cause fibrosis and vascular thrombosis.
-
-
-
Comparison of vesicular stomatitis virus pseudotyped with the S proteins from a porcine and a human coronavirus
More LessThe surface proteins S of severe acute respiratory syndrome coronavirus (SARS-CoV) and transmissible gastroenteritis virus (TGEV) were compared for their ability to mediate infection of viral pseudotypes based on vesicular stomatitis virus (VSV). The cell tropism of the respective pseudotypes corresponded to the tropism of the viruses from which the S protein was derived. Higher infectivity values were obtained with the SARS-CoV S protein than with the TGEV S protein. Differences were observed with respect to the importance of the cytoplasmic tail and the membrane anchor of the S proteins. In the case of the SARS-CoV S protein, truncation of the cytoplasmic tail resulted in increased infectivity. For the TGEV S protein, the inactivation of an intracellular retention signal in the cytoplasmic tail was required. Exchange of the membrane anchor of the S proteins led to a low infection efficiency. Our results indicate that related glycoproteins may show substantial differences in their ability to mediate pseudotype infection.
-
-
-
Interaction of severe acute respiratory syndrome-coronavirus and NL63 coronavirus spike proteins with angiotensin converting enzyme-2
More LessAlthough in different groups, the coronaviruses severe acute respiratory syndrome-coronavirus (SARS-CoV) and NL63 use the same receptor, angiotensin converting enzyme (ACE)-2, for entry into the host cell. Despite this common receptor, the consequence of entry is very different; severe respiratory distress in the case of SARS-CoV but frequently only a mild respiratory infection for NL63. Using a wholly recombinant system, we have investigated the ability of each virus receptor-binding protein, spike or S protein, to bind to ACE-2 in solution and on the cell surface. In both assays, we find that the NL63 S protein has a weaker interaction with ACE-2 than the SARS-CoV S protein, particularly in solution binding, but the residues required for contact are similar. We also confirm that the ACE-2-binding site of NL63 S lies between residues 190 and 739. A lower-affinity interaction with ACE-2 might partly explain the different pathological consequences of infection by SARS-CoV and NL63.
-
-
-
Severe acute respiratory syndrome coronavirus 3a protein activates the mitochondrial death pathway through p38 MAP kinase activation
More LessThe molecular mechanisms governing severe acute respiratory syndrome coronavirus-induced pathology are not fully understood. Virus infection and some individual viral proteins, including the 3a protein, induce apoptosis. However, the cellular targets leading to 3a protein-mediated apoptosis have not been fully characterized. This study showed that the 3a protein modulates the mitochondrial death pathway in two possible ways. Activation of caspase-8 through extrinsic signal(s) caused Bid activation. In the intrinsic pathway, there was activation of caspase-9 and cytochrome c release from the mitochondria. This was the result of increased Bax oligomerization and higher levels of p53 in 3a protein-expressing cells, which depended on the activation of p38 MAP kinase (MAPK) in these cells. For p38 activation and apoptosis induction, the 3a cytoplasmic domain was sufficient. In direct Annexin V staining assays, the 3a protein-expressing cells showed increased apoptosis that was attenuated with the p38 MAPK inhibitor SB203580. A block in nuclear translocation of the STAT3 transcription factor in cells expressing the 3a protein was also observed. These results have been used to present a model of 3a-mediated apoptosis.
-
-
-
Genomic characterizations of bat coronaviruses (1A, 1B and HKU8) and evidence for co-infections in Miniopterus bats
More LessWe previously reported the detection of bat coronaviruses (bat CoVs 1A, 1B, HKU7, HKU8 and bat-severe acute respiratory syndrome coronavirus) in Miniopterus spp. that cohabit a cave in Hong Kong. Here, we report the full genomic sequences of bat CoVs 1A, 1B and HKU8. Bat CoVs 1A and 1B, which are commonly found in the Miniopterus, are phylogenetically closely related. Using species-specific RT-PCR assays, bat CoVs 1A and 1B were confirmed to have distinct host specificities to Miniopterus magnater and Miniopterus pusillus, respectively. Interestingly, co-infections of bat CoVs 1B and HKU8 in M. pusillus are detected in seven of 38 virus-positive specimens collected from 2004 to 2006. These findings highlight that co-infections of some coronaviruses might be common events in nature. The biological basis for the host restriction of bat coronaviruses, however, is yet to be determined.
-
-
-
Identification of residues in the receptor-binding domain (RBD) of the spike protein of human coronavirus NL63 that are critical for the RBD–ACE2 receptor interaction
More LessHuman coronavirus NL63 (NL63), a member of the group I coronaviruses, may cause acute respiratory diseases in young children and immunocompromised adults. Like severe acute respiratory syndrome coronavirus (SARS-CoV), NL63 also employs the human angiotensin-converting enzyme 2 (hACE2) receptor for cellular entry. To identify residues in the spike protein of NL63 that are important for hACE2 binding, this study first generated a series of S1-truncated variants, examined their associations with the hACE2 receptor and subsequently mapped a minimal receptor-binding domain (RBD) that consisted of 141 residues (aa 476–616) towards the C terminus of the S1 domain. The data also demonstrated that the NL63 RBD bound to hACE2 more efficiently than its full-length counterpart and had a binding efficiency comparable to the S1 or RBD of SARS-CoV. A further series of RBD variants was generated using site-directed mutagenesis and random mutant library screening assays, and identified 15 residues (C497, Y498, V499, C500, K501, R518, R530, V531, G534, G537, D538, S540, E582, W585 and T591) that appeared to be critical for the RBD–hACE2 association. These critical residues clustered in three separate regions (designated RI, RII and RIII) inside the RBD, which may represent three receptor-binding sites. These results may help to delineate the molecular interactions between the S protein of NL63 and the hACE2 receptor, and may also enhance our understanding of the pathogenesis of NL63 and SARS-CoV.
-
-
-
Severe acute respiratory syndrome coronavirus Orf3a protein interacts with caveolin
More LessThe orf3a (also called X1 or U274) gene is the largest unique open reading frame in the severe acute respiratory syndrome coronavirus genome and has been proposed to encode a protein with three transmembrane domains and a large cytoplasmic domain. Recent work has suggested that the 3a protein may play a structural role in the viral life cycle, although the mechanisms for this remain uncharacterized. Here, the expression of the 3a protein in various in vitro systems is shown, it has been localized to the Golgi region and its membrane topology in transfected cells has been confirmed. Three potential caveolin-1-binding sites were reported to be present in the 3a protein. By using various biochemical, biophysical and genetic techniques, interaction of the 3a protein with caveolin-1 is demonstrated. Any one of the potential sites in the 3a protein was sufficient for this interaction. These results are discussed with respect to the possible roles of the 3a protein in the viral life cycle.
-
-
-
Comparative analysis of innate immune responses following infection of newborn calves with bovine rotavirus and bovine coronavirus
More LessBovine rotavirus (BRV) and bovine coronavirus (BCV) are important causes of diarrhoea and death in newborn calves. Although these viruses belong to distinct viral classes, they both infect intestinal epithelial cells and induce similar clinical symptoms. Rotavirus usually causes an acute infection, but coronavirus infection can persist and reoccur in adults. Differences in viral structure and clinical outcome prompted us to postulate that innate mucosal immune responses would be markedly different following rotavirus and coronavirus infections. To address this hypothesis, gene expression following BRV and BCV infection was analysed in surgically prepared intestinal loops from 1-day-old colostrum-deprived calves. Gene expression was profiled at 18 h post-infection using bovine cDNA microarrays; the majority of differentially expressed significant genes were associated with the cell cycle and innate immune responses. A select group of these genes was validated by quantitative real-time PCR (qRT-PCR). The expression of genes associated with interferons (IFNs), cytokines and Toll-like receptors, which were not present on the microarray, was analysed further by qRT-PCR. Strong activation of TLR3, IL-6 and p65 was observed in BRV-infected host tissues, but not in tissues infected with BCV. Both viruses also downregulated IFN- and pro-inflammatory cytokine-associated pathways. In vitro studies confirmed that IFN inhibited viral replication. All of these results together suggested either that very early events of host responses at 18 h post-infection were being observed, or that both viruses have unique effective strategies to evade host immune responses.
-
-
-
Persistent memory CD4+ and CD8+ T-cell responses in recovered severe acute respiratory syndrome (SARS) patients to SARS coronavirus M antigen
More LessThe membrane (M) protein of severe acute respiratory syndrome coronavirus (SARS-CoV) is a major glycoprotein with multiple biological functions. In this study, we found that memory T cells against M protein were persistent in recovered SARS patients by detecting gamma interferon (IFN-γ) production using ELISA and ELISpot assays. Flow cytometric analysis showed that both CD4+ and CD8+ T cells were involved in cellular responses to SARS-CoV M antigen. Furthermore, memory CD8+ T cells displayed an effector memory cell phenotype expressing CD45RO− CCR7− CD62L−. In contrast, the majority of IFN-γ + CD4+ T cells were central memory cells with the expression of CD45RO+ CCR7+ CD62L−. The epitope screening from 30 synthetic overlapping peptides that cover the entire SARS-CoV M protein identified four human T-cell immunodominant peptides, p21-44, p65-91, p117-140 and p200-220. All four immunodominant peptides could elicit cellular immunity with a predominance of CD8+ T-cell response. This data may have important implication for developing SARS vaccines.
-
-
-
Type I feline coronavirus spike glycoprotein fails to recognize aminopeptidase N as a functional receptor on feline cell lines
More LessThere are two types of feline coronaviruses that can be distinguished by serology and sequence analysis. Type I viruses, which are prevalent in the field but are difficult to isolate and propagate in cell culture, and type II viruses, which are less prevalent but replicate well in cell culture. An important determinant of coronavirus infection, in vivo and in cell culture, is the interaction of the virus surface glycoprotein with a cellular receptor. It is generally accepted that feline aminopeptidase N can act as a receptor for the attachment and entry of type II strains, and it has been proposed that the same molecule acts as a receptor for type I viruses. However, the experimental data are inconclusive. The aim of the studies reported here was to provide evidence for or against the involvement of feline aminopeptidase N as a receptor for type I feline coronaviruses. Our approach was to produce retroviral pseudotypes that bear the type I or type II feline coronavirus surface glycoprotein and to screen a range of feline cell lines for the expression of a functional receptor for attachment and entry. Our results show that type I feline coronavirus surface glycoprotein fails to recognize feline aminopeptidase N as a functional receptor on three continuous feline cell lines. This suggests that feline aminopeptidase N is not a receptor for type I feline coronaviruses. Our results also indicate that it should be possible to use retroviral pseudotypes to identify and characterize the cellular receptor for type I feline coronaviruses.
-
-
-
Molecular analysis of the S glycoprotein gene of bovine coronaviruses isolated in Japan from 1999 to 2006
More LessIn total, 55 isolates of Bovine coronavirus (BCoV) were collected from cases of enteric and respiratory disease occurring between 1999 and 2006 in Japan. Phylogenetic analysis of the polymorphic region of the S glycoprotein gene of these isolates, together with those of other known strains, classified the BCoV strains and isolates into four clusters. Recent field isolates display distinctive genetic divergence from the prototype enteric BCoV strains – Mebus, Quebec, Kakegawa, F15 and LY138 – and have diverged in three different aspects over 8 years. These data suggested that the genetic divergence in the polymorphic region of the S glycoprotein has progressed considerably; thus, molecular analysis of this region should be useful in investigating the molecular epidemiology of BCoV. In addition, based on the differences in amino acids among the isolates, our study did not reveal the presence of certain genetic markers of pathogenicity and clinical symptoms in this polymorphic region.
-
-
-
Full-length genome sequences of two SARS-like coronaviruses in horseshoe bats and genetic variation analysis
More LessBats were recently identified as natural reservoirs of SARS-like coronavirus (SL-CoV) or SARS coronavirus-like virus. These viruses, together with SARS coronaviruses (SARS-CoV) isolated from human and palm civet, form a distinctive cluster within the group 2 coronaviruses of the genus Coronavirus, tentatively named group 2b (G2b). In this study, complete genome sequences of two additional group 2b coronaviruses (G2b-CoVs) were determined from horseshoe bat Rhinolophus ferrumequinum (G2b-CoV Rf1) and Rhinolophus macrotis (G2b-CoV Rm1). The bat G2b-CoV isolates have an identical genome organization and share an overall genome sequence identity of 88–92 % among themselves and between them and the human/civet isolates. The most variable regions are located in the genes encoding nsp3, ORF3a, spike protein and ORF8 when bat and human/civet G2b-CoV isolates are compared. Genetic analysis demonstrated that a diverse G2b-CoV population exists in the bat habitat and has evolved from a common ancestor of SARS-CoV.
-
-
-
Inter- and intra-variant genetic heterogeneity of human coronavirus OC43 strains in France
More LessHuman coronavirus OC43 (HCoV-OC43) causes acute, self-limited respiratory infections. A close relationship between bovine coronaviruses (BCoVs) and HCoV-OC43 has recently been demonstrated. This study includes seven clinical, non-cell culture-adapted, contemporary HCoV-OC43 strains detected in France in 2003. By using RT-PCR and clonal sequencing of the S1 gene of HCoV-OC43, the inter-variant heterogeneity of the HCoV-OC43 circulating strains was studied and the intra-variant diversity was assessed by investigation of a quasispecies cloud. This paper brings to the forefront a high genetic diversity of circulating HCoV-OC43 variants. Genetically different groups are defined among the variants described in this study. One of these variants holds characteristics of an outlier and presents a deletion of 12 nt, also found in BCoV strains. Moreover, the presence of HCoV-OC43 as a quasispecies cloud in vivo during an acute respiratory-tract illness was discovered. It has also been revealed that quasispecies-cloud sizes are similar for the two viral populations tested.
-
-
-
Coronaviruses in bent-winged bats (Miniopterus spp.)
More LessA novel group 1 coronavirus was previously identified in bent-winged bats (Miniopterus spp.). Here, results are described from our ongoing surveillance of these bats for coronaviruses. These findings show that group 1 coronaviruses are endemic in these bat populations in Hong Kong. Genetic analysis of these viruses indicates that there are at least four different, but closely related, group 1 coronaviruses (bat-CoV 1A, 1B, HKU7 and HKU8) circulating in bent-winged bats. Phylogenetic analysis revealed that these group 1 bat coronaviruses have descended from a common ancestor and that these viruses have been established in these bats for a long period of time. These data provide a better understanding of the emergence and evolution of coronaviruses. Bat-CoV 1A and 1B were detected in apparently healthy Miniopterus magnater and Miniopterus pusillus, respectively, on repeated sampling occasions at a single habitat, suggesting that these viruses have established a persistent infection in these populations.
-
-
-
Interaction of severe acute respiratory syndrome-associated coronavirus with dendritic cells
More LessSevere acute respiratory syndrome (SARS) of humans is caused by a novel coronavirus of zoonotic origin termed SARS-associated coronavirus (SARS-CoV). The virus induces severe injury of lung tissue, as well as lymphopenia and destruction of the architecture of lymphatic tissue by as-yet-unknown mechanisms. In this study, the interaction of SARS-CoV with dendritic cells (DCs), the key regulators of immune responses, was analysed. Monocyte-derived DCs were infected with SARS-CoV and analysed for viability, surface-marker expression and alpha interferon (IFN-α) induction. SARS-CoV infection was monitored by quantitative RT-PCR, immunofluorescence analysis and recovery experiments. SARS-CoV infected both immature and mature DCs, although replication efficiency was low. Immature DCs were activated by SARS-CoV infection and by UV-inactivated SARS-CoV. Infected DCs were still viable on day 6 post-infection, but major histocompatibility complex class I upregulation was missing, indicating that DC function was impaired. Additionally, SARS-CoV infection induced a delayed activation of IFN-α expression. Therefore, it is concluded that SARS-CoV has the ability to circumvent both the innate and the adaptive immune systems.
-
-
-
Analysis of ACE2 in polarized epithelial cells: surface expression and function as receptor for severe acute respiratory syndrome-associated coronavirus
More LessThe primary target of severe acute respiratory syndrome-associated coronavirus (SARS-CoV) is epithelial cells in the respiratory and intestinal tract. The cellular receptor for SARS-CoV, angiotensin-converting enzyme 2 (ACE2), has been shown to be localized on the apical plasma membrane of polarized respiratory epithelial cells and to mediate infection from the apical side of these cells. Here, these results were confirmed and extended by including a colon carcinoma cell line (Caco-2), a lung carcinoma cell line (Calu-3) and Vero E6 cells in our analysis. All three cell types expressed human ACE2 on the apical membrane domain and were infected via this route, as determined with vesicular stomatitis virus pseudotypes containing the S protein of SARS-CoV. In a histological analysis of the respiratory tract, ACE2 was detected in the trachea, main bronchus and alveoli, and occasionally also in the small bronchi. These data will help us to understand the pathogenesis of SARS-CoV infection.
-
-
-
Analysis of human coronavirus 229E spike and nucleoprotein genes demonstrates genetic drift between chronologically distinct strains
More LessHistorically, coronaviruses have been recognized as a cause of minor respiratory infections in humans. However, the recent identification of three novel human coronaviruses, one causing severe acute respiratory syndrome (SARS), has prompted further examination of these viruses. Previous studies of geographically and chronologically distinct Human coronavirus 229E (HCoV-229E) isolates have found only limited variation within S gene nucleotide sequences. In contrast, analysis of the S genes of contemporary Human coronavirus OC43 variants identified in Belgium revealed two distinct viruses circulating during 2003 and 2004. Here, the S and N gene sequences of 25 HCoV-229E variants identified in Victoria, Australia, between 1979 and 2004 in patients with symptomatic infections were determined. Phylogenetic analysis showed clustering of the isolates into four groups, with evidence of increasing divergence with time. Evidence of positive selection in the S gene was also established.
-
-
-
Comparative evaluation of two severe acute respiratory syndrome (SARS) vaccine candidates in mice challenged with SARS coronavirus
More LessRaymond H. See, Alexander N. Zakhartchouk, Martin Petric, David J. Lawrence, Catherine P. Y. Mok, Robert J. Hogan, Thomas Rowe, Lois A. Zitzow, Karuna P. Karunakaran, Mary M. Hitt, Frank L. Graham, Ludvik Prevec, James B. Mahony, Chetna Sharon, Thierry C. Auperin, James M. Rini, Aubrey J. Tingle, David W. Scheifele, Danuta M. Skowronski, David M. Patrick, Thomas G. Voss, Lorne A. Babiuk, Jack Gauldie, Rachel L. Roper, Robert C. Brunham and B. Brett FinlayTwo different severe acute respiratory syndrome (SARS) vaccine strategies were evaluated for their ability to protect against live SARS coronavirus (CoV) challenge in a murine model of infection. A whole killed (inactivated by β-propiolactone) SARS-CoV vaccine and a combination of two adenovirus-based vectors, one expressing the nucleocapsid (N) and the other expressing the spike (S) protein (collectively designated Ad S/N), were evaluated for the induction of serum neutralizing antibodies and cellular immune responses and their ability to protect against pulmonary SARS-CoV replication. The whole killed virus (WKV) vaccine given subcutaneously to 129S6/SvEv mice was more effective than the Ad S/N vaccine administered either intranasally or intramuscularly in inhibiting SARS-CoV replication in the murine respiratory tract. This protective ability of the WKV vaccine correlated with the induction of high serum neutralizing-antibody titres, but not with cellular immune responses as measured by gamma interferon secretion by mouse splenocytes. Titres of serum neutralizing antibodies induced by the Ad S/N vaccine administered intranasally or intramuscularly were significantly lower than those induced by the WKV vaccine. However, Ad S/N administered intranasally, but not intramuscularly, significantly limited SARS-CoV replication in the lungs. Among the vaccine groups, SARS-CoV-specific IgA was found only in the sera of mice immunized intranasally with Ad S/N, suggesting that mucosal immunity may play a role in protection for the intranasal Ad S/N delivery system. Finally, the sera of vaccinated mice contained antibodies to S, further suggesting a role for this protein in conferring protective immunity against SARS-CoV infection.
-
-
-
Amino terminus of the SARS coronavirus protein 3a elicits strong, potentially protective humoral responses in infected patients
More LessThe 3a protein of severe acute respiratory syndrome (SARS)-associated coronavirus is expressed and transported to the plasma membrane in tissue cells of infected patients. Its short N-terminal ectodomain was found to elicit strong humoral responses in half of the patients who had recovered from SARS. The ectodomain-specific antibodies from the convalescent-phase plasma readily recognized and induced destruction of 3a-expressing cells in the presence of the human complement system, demonstrating their potential ability to provide immune protection by recognizing and eliminating SARS coronavirus-infected cells that express the target protein. In addition, when coupled to a carrier protein, the ectodomain peptide elicited 3a-specific antibodies in mice and rabbit at high titres. These results showed that the N terminus of the 3a protein is highly immunogenic and elicits potentially protective humoral responses in infected patients. Therefore, the short extracellular domain may be a valuable immunogen in the development of a vaccine for infectious SARS.
-
-
-
Subcellular localization of the severe acute respiratory syndrome coronavirus nucleocapsid protein
More LessThe coronavirus nucleocapsid (N) protein is a viral RNA-binding protein with multiple functions in terms of virus replication and modulating cell signalling pathways. N protein is composed of three distinct regions containing RNA-binding motif(s), and appropriate signals for modulating cell signalling. The subcellular localization of severe acute respiratory syndrome coronavirus (SARS-CoV) N protein was studied. In infected cells, SARS-CoV N protein localized exclusively to the cytoplasm. In contrast to the avian coronavirus N protein, overexpressed SARS-CoV N protein remained principally localized to the cytoplasm, with very few cells exhibiting nucleolar localization. Bioinformatic analysis and deletion mutagenesis coupled to confocal microscopy and live-cell imaging, revealed that SARS-CoV N protein regions I and III contained nuclear localization signals and region II contained a nucleolar retention signal. However, cytoplasmic localization was directed by region III and was the dominant localization signal in the protein.
-
-
-
Genomic RNA sequence of Feline coronavirus strain FIPV WSU-79/1146
More LessA consensus sequence of the Feline coronavirus (FCoV) (strain FIPV WSU-79/1146) genome was determined from overlapping cDNA fragments produced by RT-PCR amplification of viral RNA. The genome was found to be 29 125 nt in length, excluding the poly(A) tail. Analysis of the sequence identified conserved open reading frames and revealed an overall genome organization similar to that of other coronaviruses. The genomic RNA was analysed for putative cis-acting elements and the pattern of subgenomic mRNA synthesis was analysed by Northern blotting. Comparative sequence analysis of the predicted FCoV proteins identified 16 replicase proteins (nsp1–nsp16) and four structural proteins (spike, membrane, envelope and nucleocapsid). Two mRNAs encoding putative accessory proteins were also detected. Phylogenetic analyses confirmed that FIPV WSU-79/1146 belongs to the coronavirus subgroup G1-1. These results confirm and extend previous findings from partial sequence analysis of FCoV genomes.
-
-
-
Vesicular stomatitis virus pseudotyped with severe acute respiratory syndrome coronavirus spike protein
More LessSevere acute respiratory syndrome coronavirus (SARS-CoV) contains a single spike (S) protein, which binds to its receptor, angiotensin-converting enzyme 2 (ACE2), induces membrane fusion and serves as a neutralizing antigen. A SARS-CoV-S protein-bearing vesicular stomatitis virus (VSV) pseudotype using the VSVΔG* system was generated. Partial deletion of the SARS-CoV-S protein cytoplasmic domain allowed efficient incorporation into VSV particles and led to the generation of a pseudotype (VSV-SARS-St19) at high titre. Green fluorescent protein expression was demonstrated as early as 7 h after infection of Vero E6 cells with VSV-SARS-St19. VSV-SARS-St19 was neutralized by anti-SARS-CoV antibody and soluble ACE2, and its infection was blocked by treatment of Vero E6 cells with anti-ACE2 antibody. These results indicated that VSV-SARS-St19 infection is mediated by SARS-CoV-S protein in an ACE2-dependent manner. VSV-SARS-St19 will be useful for analysing the function of SARS-CoV-S protein and for developing rapid methods of detecting neutralizing antibodies specific for SARS-CoV infection.
-
-
-
The 3a protein of severe acute respiratory syndrome-associated coronavirus induces apoptosis in Vero E6 cells
More LessAn outbreak of severe acute respiratory syndrome (SARS) occurred in China and the first case emerged in mid-November 2002. The aetiological agent of this disease was found to be a previously unknown coronavirus, SARS-associated coronavirus (SARS-CoV). The detailed pathology of SARS-CoV infection and the host response to the viral infection are still not known. The 3a gene encodes a non-structural viral protein, which is predicted to be a transmembrane protein. In this study, it was shown that the 3a protein was expressed in the lungs and intestinal tissues of SARS patients and that the protein localized to the endoplasmic reticulum in 3a-transfected monkey kidney Vero E6 cells. In vitro experiments of chromatin condensation and DNA fragmentation suggested that the 3a protein may trigger apoptosis. These data showed that overexpression of a single SARS-CoV protein can induce apoptosis in vitro.
-
-
-
Molecular identification and characterization of novel coronaviruses infecting graylag geese (Anser anser), feral pigeons (Columbia livia) and mallards (Anas platyrhynchos)
More LessIn light of the finding of a previously unknown coronavirus as the aetiology of the severe acute respiratory syndrome (SARS), it is probable that other coronaviruses, than those recognized to date, are circulating in animal populations. Here, the results of a screening for coronavirus are presented, using a universal coronavirus RT-PCR, of the bird species graylag goose (Anser anser), feral pigeon (Columbia livia) and mallard (Anas platyrhynchos). Coronaviruses were found in cloacal swab samples from all the three bird species. In the graylag goose, 40 of 163 sampled birds were coronavirus positive, whereas two of 100 sampled pigeons and one of five sampled mallards tested positive. The infected graylag geese showed lower body weights compared with virus-negative birds, suggesting clinical significance of the infection. Phylogenetic analyses performed on the replicase gene and nucleocapsid protein sequences, indicated that the novel coronaviruses described in the present study all branch off from group III coronaviruses. All the novel avian coronaviruses harboured the conserved s2m RNA structure in their 3′ untranslated region, like other previously described group III coronaviruses, and like the SARS coronavirus. Sequencing of the complete nucleocapsid gene and downstream regions of goose and pigeon coronaviruses, evidenced the presence of two additional open reading frames for the goose coronavirus with no sequence similarity to known proteins, but with predicted transmembrane domains for one of the encoded proteins, and one additional open reading frame for the pigeon coronavirus, with a predicted transmembrane domain, downstream of the nucleocapsid gene.
-
-
-
A single immunization with a rhabdovirus-based vector expressing severe acute respiratory syndrome coronavirus (SARS-CoV) S protein results in the production of high levels of SARS-CoV-neutralizing antibodies
More LessForeign viral proteins expressed by rabies virus (RV) have been shown to induce potent humoral and cellular immune responses in immunized animals. In addition, highly attenuated and, therefore, very safe RV-based vectors have been constructed. Here, an RV-based vaccine vehicle was utilized as a novel vaccine against severe acute respiratory syndrome coronavirus (SARS-CoV). For this approach, the SARS-CoV nucleocapsid protein (N) or envelope spike protein (S) genes were cloned between the RV glycoprotein G and polymerase L genes. Recombinant vectors expressing SARS-CoV N or S protein were recovered and their immunogenicity was studied in mice. A single inoculation with the RV-based vaccine expressing SARS-CoV S protein induced a strong SARS-CoV-neutralizing antibody response. The ability of the RV-SARS-CoV S vector to confer immunity after a single inoculation makes this live vaccine a promising candidate for eradication of SARS-CoV in animal reservoirs, thereby reducing the risk of transmitting the infection to humans.
-
-
-
Differential maturation and subcellular localization of severe acute respiratory syndrome coronavirus surface proteins S, M and E
More LessPost-translational modifications and correct subcellular localization of viral structural proteins are prerequisites for assembly and budding of enveloped viruses. Coronaviruses, like the severe acute respiratory syndrome-associated virus (SARS-CoV), bud from the endoplasmic reticulum-Golgi intermediate compartment. In this study, the subcellular distribution and maturation of SARS-CoV surface proteins S, M and E were analysed by using C-terminally tagged proteins. As early as 30 min post-entry into the endoplasmic reticulum, high-mannosylated S assembles into trimers prior to acquisition of complex N-glycans in the Golgi. Like S, M acquires high-mannose N-glycans that are subsequently modified into complex N-glycans in the Golgi. The N-glycosylation profile and the absence of O-glycosylation on M protein relate SARS-CoV to the previously described group 1 and 3 coronaviruses. Immunofluorescence analysis shows that S is detected in several compartments along the secretory pathway from the endoplasmic reticulum to the plasma membrane while M predominantly localizes in the Golgi, where it accumulates, and in trafficking vesicles. The E protein is not glycosylated. Pulse-chase labelling and confocal microscopy in the presence of protein translation inhibitor cycloheximide revealed that the E protein has a short half-life of 30 min. E protein is found in bright perinuclear patches colocalizing with endoplasmic reticulum markers. In conclusion, SARS-CoV surface proteins S, M and E show differential subcellular localizations when expressed alone suggesting that additional cellular or viral factors might be required for coordinated trafficking to the virus assembly site in the endoplasmic reticulum-Golgi intermediate compartment.
-
-
-
Isolation of avian infectious bronchitis coronavirus from domestic peafowl (Pavo cristatus) and teal (Anas)
More LessCoronavirus-like viruses, designated peafowl/China/LKQ3/2003 (pf/CH/LKQ3/03) and teal/China/LDT3/2003 (tl/CH/LDT3/03), were isolated from a peafowl and a teal during virological surveillance in Guangdong province, China. Partial genomic sequence analysis showed that these isolates had the S–3–M–5–N gene order that is typical of avian coronaviruses. The spike, membrane and nucleocapsid protein genes of pf/CH/LKQ3/03 had >99 % identity to those of the avian infectious bronchitis coronavirus H120 vaccine strain (Massachusetts serotype) and other Massachusetts serotype isolates. Furthermore, when pf/CH/LKQ3/03 was inoculated experimentally into chickens (specific-pathogen-free), no disease signs were apparent. tl/CH/LDT3/03 had a spike protein gene with 95 % identity to that of a Chinese infectious bronchitis virus (IBV) isolate, although more extensive sequencing revealed the possibility that this strain may have undergone recombination. When inoculated into chickens, tl/CH/LDT3/03 resulted in the death of birds from nephritis. Taken together, this information suggests that pf/CH/LKQ3/03 might be a revertant, attenuated vaccine IBV strain, whereas tl/CH/LDT3/03 is a nephropathogenic field IBV strain, generated through recombination. The replication and non-pathogenic nature of IBV in domestic peafowl and teal under field conditions raises questions as to the role of these hosts as carriers of IBV and the potential that they may have to transmit virus to susceptible chicken populations.
-
-
-
Severe acute respiratory syndrome coronavirus nucleocapsid protein expressed by an adenovirus vector is phosphorylated and immunogenic in mice
More LessSevere acute respiratory syndrome coronavirus (SARS-CoV) has been identified as the aetiological agent of SARS. Thus, vaccination against SARS-CoV may represent an effective approach towards controlling SARS. The nucleocapsid (N) protein is thought to play a role in induction of cell-mediated immunity to SARS-CoV and thus it is important to characterize this protein. In the present study, an E1/partially E3-deleted, replication-defective human adenovirus 5 (Ad5) vector (Ad5-N-V) expressing the SARS-CoV N protein was constructed. The N protein, expressed in vitro by Ad5-N-V, was of the expected molecular mass of 50 kDa and was phosphorylated. Vaccination of C57BL/6 mice with Ad5-N-V generated potent SARS-CoV-specific humoral and T cell-mediated immune responses. These results show that Ad5-N-V may potentially be used as a SARS-CoV vaccine.
-
-
-
Rapid identification of coronavirus replicase inhibitors using a selectable replicon RNA
More LessA previously unknown coronavirus (CoV) is the aetiological agent causing severe acute respiratory syndrome (SARS), for which an effective antiviral treatment is urgently needed. To enable the rapid and biosafe identification of coronavirus replicase inhibitors, we have generated a non-cytopathic, selectable replicon RNA (based on human CoV 229E) that can be stably maintained in eukaryotic cells. Most importantly, the replicon RNA mediates reporter gene expression as a marker for coronavirus replication. We have used a replicon RNA-containing cell line to test the inhibitory effect of several compounds that are currently being assessed for SARS treatment. Amongst those, interferon-α displayed the strongest inhibitory activity. Our results demonstrate that coronavirus replicon cell lines provide a versatile and safe assay for the identification of coronavirus replicase inhibitors. Once this technology is adapted to SARS-CoV replicon RNAs, it will allow high throughput screening for SARS-CoV replicase inhibitors without the need to grow infectious SARS-CoV.
-
-
-
Antibody response and viraemia during the course of severe acute respiratory syndrome (SARS)-associated coronavirus infection
More LessTo understand the time-course of viraemia and antibody responses to severe acute respiratory syndrome-associated coronavirus (SARS-CoV), RT-PCR and ELISA were used to assay 376 blood samples from 135 SARS patients at various stages of the illness, including samples from patients who were in their early convalescent phase. The results showed that IgM antibodies decreased and became undetectable 11 weeks into the recovery phase. IgG antibodies, however, remained detectable for a period beyond 11 weeks and were found in 100 % of patients in the early convalescent phase. SARS-CoV viraemia mainly appeared 1 week after the onset of illness and then decreased over a period of 1 month, becoming undetectable in the blood samples of the convalescent patients. At the peak of viraemia, viral RNA was detectable in 75 % of blood samples from patients who were clinically diagnosed with SARS 1 or 2 weeks before the test.
-
-
-
Proliferative growth of SARS coronavirus in Vero E6 cells
More LessAn isolate of SARS coronavirus (strain 2003VA2774) was obtained from a patient and used to infect Vero E6 cells. The replication cycle of the virus was followed from 1 to 30 h post-infection (p.i.). It was surprising to observe the swift growth of this human virus in Vero cells. Within the first hour of infection, the most obvious ultrastructural change was the proliferation of the Golgi complexes and related vesicles accompanied by swelling of some of the trans-Golgi sacs. Extracellular virus particles were present by 5 h p.i. in about 5 % of the cells and this increased dramatically to about 30 % of the cell population within an hour (6 h p.i.). Swollen Golgi sacs contained virus nucleocapsids at different stages of maturation. These virus precursors were also in large vacuoles and in close association with membrane whorls. The membrane whorls could be the replication complexes, since they appeared rather early in the replication cycle. As infection progressed from 12 to 21 h p.i., the cytoplasm of the infected cells was filled with numerous large, smooth-membraned vacuoles containing a mixture of mature virus and spherical cores. Several of these vacuoles were close to the cell periphery, ready to export out the mature progeny virus particles via exocytosis. By 24 to 30 h p.i., crystalline arrays of the extracellular virus particles were seen commonly at the cell surface.
-
-
-
Persistence and transmission of natural type I feline coronavirus infection
More LessTo examine the mode of natural transmission and persistence of feline coronavirus (FCoV), FCoV strains shed by domestic cats were investigated over periods of up to 7 years. An RT-PCR that amplified part of the 3′ end of the viral spike (S) gene was devised to distinguish FCoV types I and II. All but 1 of 28 strains of FCoV from 43 cats were type I. Nucleotide identities of the amplified 320 bp product from 49 type I FCoVs ranged from 79 to 100 %. The consensus partial S sequence of isolates recovered from persistently infected cats at time intervals spanning years was generally conserved. While most cats were infected with a single strain, a few may have been infected by more than one strain. Cats that were transiently infected and ceased shedding could be re-infected with either the same, or a different, strain. In most cases, whether a cat became persistently or transiently infected was independent of the virus strain. However, one strain was unusual in that it infected the majority of cats in the household simultaneously and was still being shed 18 months later. Factors that influence whether FCoV establishes lifelong infection in some cats and not others are determined mainly by the host response to infection.
-
-
-
Mechanisms and enzymes involved in SARS coronavirus genome expression
More LessA novel coronavirus is the causative agent of the current epidemic of severe acute respiratory syndrome (SARS). Coronaviruses are exceptionally large RNA viruses and employ complex regulatory mechanisms to express their genomes. Here, we determined the sequence of SARS coronavirus (SARS-CoV), isolate Frankfurt 1, and characterized key RNA elements and protein functions involved in viral genome expression. Important regulatory mechanisms, such as the (discontinuous) synthesis of eight subgenomic mRNAs, ribosomal frameshifting and post-translational proteolytic processing, were addressed. Activities of three SARS coronavirus enzymes, the helicase and two cysteine proteinases, which are known to be critically involved in replication, transcription and/or post-translational polyprotein processing, were characterized. The availability of recombinant forms of key replicative enzymes of SARS coronavirus should pave the way for high-throughput screening approaches to identify candidate inhibitors in compound libraries.
-
-
-
Sequence of the 3′-terminal end (8·1 kb) of the genome of porcine haemagglutinating encephalomyelitis virus: comparison with other haemagglutinating coronaviruses
More LessA cytopathogenic coronavirus, serologically identified as porcine haemagglutinating encephalomyelitis virus (HEV), has recently been associated with acute outbreaks of wasting and encephalitis in nursing piglets from pig farms in southern Québec and Ontario, Canada. The 3′-terminal end of the genome of the prototype HEV-67N strain and that of the recent Québec IAF-404 field isolate, both propagated in HRT-18 cells, were sequenced. Overall, sequencing data indicated that HEV has remained antigenically and genetically stable since its first isolation in North America in 1962. Compared with the prototype strain of bovine enteropathogenic coronavirus (BCoV), HEV, as well as the human respiratory coronavirus (HCoV-OC43) showed a major deletion in their ORF4 gene. Deduced amino acid sequences for both HEV strains revealed 89/88, 80, 93/92 and 95/94% identities with the structural proteins HE, S, M and N of BCoV and HCoV-OC43, respectively. Major variations were observed in the S1 portion of the S gene of both HEV strains, with only 73/71% amino acid identities compared with those of the two other haemagglutinating coronaviruses.
-
-
-
Conservation of substrate specificities among coronavirus main proteases
More LessThe key enzyme in coronavirus replicase polyprotein processing is the coronavirus main protease, 3CLpro. The substrate specificities of five coronavirus main proteases, including the prototypic enzymes from the coronavirus groups I, II and III, were characterized. Recombinant main proteases of human coronavirus (HCoV), transmissible gastroenteritis virus (TGEV), feline infectious peritonitis virus, avian infectious bronchitis virus and mouse hepatitis virus (MHV) were tested in peptide-based trans-cleavage assays. The determination of relative rate constants for a set of corresponding HCoV, TGEV and MHV 3CLpro cleavage sites revealed a conserved ranking of these sites. Furthermore, a synthetic peptide representing the N-terminal HCoV 3CLpro cleavage site was shown to be effectively hydrolysed by noncognate main proteases. The data show that the differential cleavage kinetics of sites within pp1a/pp1ab are a conserved feature of coronavirus main proteases and lead us to predict similar processing kinetics for the replicase polyproteins of all coronaviruses.
-
-
-
Mutational analysis of the active centre of coronavirus 3C-like proteases
More LessFormation of the coronavirus replication–transcription complex involves the synthesis of large polyprotein precursors that are extensively processed by virus-encoded cysteine proteases. In this study, the coding sequence of the feline infectious peritonitis virus (FIPV) main protease, 3CLpro, was determined. Comparative sequence analyses revealed that FIPV 3CLpro and other coronavirus main proteases are related most closely to the 3C-like proteases of potyviruses. The predicted active centre of the coronavirus enzymes has accepted unique replacements that were probed by extensive mutational analysis. The wild-type FIPV 3CLpro domain and 25 mutants were expressed in Escherichia coli and tested for proteolytic activity in a peptide-based assay. The data strongly suggest that, first, the FIPV 3CLpro catalytic system employs His41 and Cys144 as the principal catalytic residues. Second, the amino acids Tyr160 and His162, which are part of the conserved sequence signature Tyr160–Met161–His162 and are believed to be involved in substrate recognition, were found to be indispensable for proteolytic activity. Third, replacements of Gly83 and Asn64, which were candidates to occupy the position spatially equivalent to that of the catalytic Asp residue of chymotrypsin-like proteases, resulted in proteolytically active proteins. Surprisingly, some of the Asn64 mutants even exhibited strongly increased activities. Similar results were obtained for human coronavirus (HCoV) 3CLpro mutants in which the equivalent Asn residue (HCoV 3CLpro Asn64) was substituted. These data lead us to conclude that both the catalytic systems and substrate-binding pockets of coronavirus main proteases differ from those of other RNA virus 3C and 3C-like proteases.
-
-
-
The sialate-4-O-acetylesterases of coronaviruses related to mouse hepatitis virus: a proposal to reorganize group 2 Coronaviridae
More LessGroup 2 coronaviruses are characterized within the order Nidovirales by a unique genome organization. A characteristic feature of group 2 coronaviruses is the presence of a gene encoding the haemagglutinin–esterase (HE) protein, which is absent in coronaviruses of groups 1 and 3. At least three coronavirus strains within group 2 expressed a structural protein with sialate-4-O-acetylesterase activity, distinguishing them from other members of group 2, which encode an enzyme specific for 5-N-acetyl-9-O-acetylneuraminic acid. The esterases of mouse hepatitis virus (MHV) strains S and JHM and puffinosis virus (PV) specifically hydrolysed 5-N-acetyl-4-O-acetylneuraminic acid (Neu4,5Ac2) as well as the synthetic substrates p-nitrophenyl acetate, 4-methylumbelliferyl acetate and fluorescein diacetate. The K m values of the MHV-like esterases for the latter substrates were two- to tenfold lower than those of the sialate-9-O-acetylesterases of influenza C viruses. Another unspecific esterase substrate, α-naphthyl acetate, was used for the in situ detection of the dimeric HE proteins in SDS–polyacrylamide gels. MHV-S, MHV-JHM and PV bound to horse serum glycoproteins containing Neu4,5Ac2. De-O-acetylation of the glycoproteins by alkaline treatment or incubation with the viral esterases resulted in a complete loss of recognition, indicating a specific interaction of MHV-like coronaviruses with Neu4,5Ac2. Combined with evidence for distinct phylogenetic lineages of group 2 coronaviruses, subdivision into subgroups 2a (MHV-like viruses) and 2b (bovine coronavirus-like viruses) is suggested.
-
-
-
Comparison of genomic and predicted amino acid sequences of respiratory and enteric bovine coronaviruses isolated from the same animal with fatal shipping pneumonia
More LessThe complete genome sequences are reported here of two field isolates of bovine coronavirus (BCoV), which were isolated from respiratory and intestinal samples of the same animal experiencing fatal pneumonia during a bovine shipping fever epizootic. Both genomes contained 31028 nucleotides and included 13 open reading frames (ORFs) flanked by 5′- and 3′-untranslated regions (UTRs). ORF1a and ORF1b encode replicative polyproteins pp1a and pp1ab, respectively, that contain all of the putative functional domains documented previously for the closest relative, mouse hepatitis virus. The genomes of the BCoV isolates differed in 107 positions, scattered throughout the genome except the 5′-UTR. Differences in 25 positions were non-synonymous and were located in all proteins except pp1b. Six replicase mutations were identified within or immediately downstream of the predicted largest pp1a-derived protein, p195/p210. Single amino acid changes within p195/p210 as well as within the S glycoprotein might contribute to the different phenotypes of the BCoV isolates.
-
-
-
Infectious RNA transcribed in vitro from a cDNA copy of the human coronavirus genome cloned in vaccinia virus
More LessThe coronavirus genome is a positive-strand RNA of extraordinary size and complexity. It is composed of approximately 30000 nucleotides and it is the largest known autonomously replicating RNA. It is also remarkable in that more than two-thirds of the genome is devoted to encoding proteins involved in the replication and transcription of viral RNA. Here, a reverse-genetic system is described for the generation of recombinant coronaviruses. This system is based upon the in vitro transcription of infectious RNA from a cDNA copy of the human coronavirus 229E genome that has been cloned and propagated in vaccinia virus. This system is expected to provide new insights into the molecular biology and pathogenesis of coronaviruses and to serve as a paradigm for the genetic analysis of large RNA virus genomes. It also provides a starting point for the development of a new class of eukaryotic, multi-gene RNA vectors that are able to express several proteins simultaneously.
-
-
-
Functional analysis of an epitope in the S2 subunit of the murine coronavirus spike protein: involvement in fusion activity
More LessThe monoclonal antibody (MAb) 5B19.2, which has virus-neutralizing and fusion inhibition activities, binds to an epitope (S2A) consisting of nine hydrophobic amino acids in the S2 subunit of the mouse hepatitis virus (MHV) spike (S) protein. This suggests that the S2A epitope may be involved in binding the virus to the MHV receptor and/or in virus–cell fusion. Co-immunoprecipitation analyses demonstrated that while the binding of virus to the receptor was blocked by anti-S1 MAbs, it was not blocked by the S2A antiserum, indicating that S2A was not involved in receptor-binding. The S proteins prepared in this study with mutations in the S2A epitope were either fusogenic or non-fusogenic and their fusogenicity did not correlate with the hydrophobic feature of the S2A epitope. All of these wt and mutated S proteins were similarly transported onto the cell membrane independent of their fusogenicity capability. These results suggest that S2A may mediate the fusion activity of the MHV S protein during virus entry into cells.
-
-
-
Temperature-sensitive acetylesterase activity of haemagglutinin-esterase specified by respiratory bovine coronaviruses
More LessNumerous respiratory bovine coronaviruses (RBCV) were isolated recently from nasal swab samples and lung tissues of feedlot cattle with acute respiratory tract disease. These newly emerging RBCV isolates exhibited distinct phenotypic features that differentiated them from enteropathogenic bovine coronaviruses (EBCV). The RBCV strains had a receptor-destroying enzyme function mediated by acetylesterase (AE) activity of the haemagglutinin-esterase (HE) glycoprotein. The HE genes of wild-type EBCV strain LY138 and RBCV strains OK-0514 (OK) and LSU-94LSS-051 (LSU) were cloned, sequenced and transiently expressed in COS-7 cells. The enzymic properties of HE proteins in COS-7 cellular extracts and in purified virus preparations were assayed at room temperature, 37°C and 39°C by two different assays. One assay used ρ-nitrophenyl acetate (PNPA) as substrate and detected serine-esterase activity; the second assay monitored AE function with bovine submaxillary mucin (BSM) as substrate. The PNPA tests confirmed that HE proteins of EBCV and RBCV were functionally expressed in transfected COS-7 cells. Time-dependent determination of the AE activity of purified RBCV OK and LSU particles showed lower AE activity at 39°C than at 37°C, whereas the purified EBCV LY particles retained full AE activity at both 37°C and 39°C. Transiently expressed RBCV HE exhibited a marked reduction of AE activity after 40 min of assay time at 37°C. In contrast, the AE activity of the transiently expressed EBCV HE remained stable beyond 40 min. The deduced amino-acid sequences of the HE proteins specified by the RBCV strains OK and LSU contained specific amino-acid changes in comparison with the EBCV LY strain, which may be responsible for the observed enzymic differences. These results are consistent with the hypothesis that RBCV strains have evolved to selectively replicate in respiratory tissues and that HE may play a role in this tissue tropism.
-
-
-
Utilizing fowlpox virus recombinants to generate defective RNAs of the coronavirus infectious bronchitis virus
More LessCoronavirus defective RNAs (D-RNAs) have been used as RNA vectors for the expression of heterologous genes and as vehicles for reverse genetics by modifying coronavirus genomes by targetted recombination. D-RNAs based on the avian coronavirus infectious bronchitis virus (IBV) D-RNA CD-61 have been rescued (replicated and packaged into virions) in a helper virus-dependent manner following electroporation of in vitro-generated T7 transcripts into IBV-infected cells. In order to increase the efficiency of rescue of IBV D-RNAs, cDNAs based on CD-61, under the control of a T7 promoter, were integrated into the fowlpox virus (FPV) genome. The 3′-UTR of the D-RNAs was flanked by a hepatitis delta antigenomic ribozyme and T7 terminator sequence to generate suitable 3′ ends for rescue by helper IBV. Cells were co-infected simultaneously with IBV, the recombinant FPV (rFPV) containing the D-RNA sequence and a second rFPV expressing T7 RNA polymerase for the initial expression of the D-RNA transcript, subsequently rescued by helper IBV. Rescue of rFPV-derived CD-61 occurred earlier and with higher efficiency than demonstrated previously for electroporation of in vitro T7-generated RNA transcripts in avian cells. Rescue of CD-61 was also demonstrated for the first time in mammalian cells. The rescue of rFPV-derived CD-61 by M41 helper IBV resulted in leader switching, in which the Beaudette-type leader sequence on CD-61 was replaced with the M41 leader sequence, confirming that helper IBV virus replicated the rFPV-derived D-RNA. An rFPV-derived D-RNA containing the luciferase gene under the control of an IBV transcription-associated sequence was also rescued and expressed luciferase on serial passage.
-
-
-
A potential role for tumour necrosis factor-α in synergy between porcine respiratory coronavirus and bacterial lipopolysaccharide in the induction of respiratory disease in pigs
More LessThis study examined whether exposure of pigs to both porcine respiratory coronavirus (PRCV) and bacterial lipopolysaccharide (LPS) can potentiate respiratory disease and lung secretion of tumour necrosis factor-α (TNF-α) and interleukin-1 (IL-1). Caesarian-derived colostrum-deprived pigs were inoculated intratracheally with PRCV, with LPS from Escherichia coli O111:B4 (20 μg/kg), or with a combination of the two, and killed at set times after inoculation. Clinical signs, virus replication and (histo)pathological changes in the lungs, percentage of neutrophils and bioactive TNF-α and IL-1 in broncho-alveolar lavage (BAL) fluids were examined. The effects of separate virus or LPS inoculations were subclinical and failed to induce high and sustained cytokine levels. In a preliminary study, pigs were inoculated with PRCV and then with LPS 24 h later and killed sequentially. Severe respiratory disease and significantly enhanced TNF-α titres (208–3601 U/ml versus 40–89 U/ml after LPS only) were seen during the first 12 h after LPS inoculation. IL-1 levels (106–1631 U/ml versus 28–654 U/ml after LPS only) were also increased, but persisted for longer after clinical recovery than TNF-α. In a second study, pigs were inoculated with PRCV and subsequently with LPS at various time intervals ranging from 0 to 24 h, and killed 5 h after inoculation with LPS. A time interval of at least 12 h between inoculations was necessary for prominent respiratory signs to develop. Production of TNF-α, but not IL-1, was also dependent on the time interval between inoculations and was tightly correlated with disease. Lung neutrophil infiltration and pathological changes were comparable after combined PRCV-LPS and single LPS inoculations, and were not associated with disease. These data show that exposure to high endotoxin concentrations in swine buildings can precipitate respiratory disease in PRCV-infected pigs, and that TNF-α is probably an important mediator of these effects. This is the first in-vivo demonstration of synergy between respiratory viruses and LPS.
-
-
-
Expression of reporter genes from the defective RNA CD-61 of the coronavirus infectious bronchitis virus
More LessThe defective RNA (D-RNA) CD-61, derived from the Beaudette strain of the avian coronavirus infectious bronchitis virus (IBV), was used as an RNA vector for the expression of two reporter genes, luciferase and chloramphenicol acetyltransferase (CAT). D-RNAs expressing the CAT gene were demonstrated to be capable of producing CAT protein in a helper-dependent expression system to about 1·6 μg per 106 cells. The reporter genes were expressed from two different sites within the CD-61 sequence and expression was not affected by interruption of the CD-61-specific ORF. Expression of the reporter genes was under the control of a transcription-associated sequence (TAS) derived from the Beaudette gene 5, normally used for the transcription of IBV subgenomic mRNA 5. The Beaudette gene 5 TAS is composed of two tandem repeats of the IBV canonical consensus sequence involved in the acquisition of a leader sequence during the discontinuous transcription of IBV subgenomic mRNAs. It is demonstrated that only one canonical sequence is required for expression of mRNA 5 or for the expression of an mRNA from a D-RNA and that either sequence can function as an acceptor site for acquisition of the leader sequence.
-
-
-
Gill-associated virus of Penaeus monodon prawns: an invertebrate virus with ORF1a and ORF1b genes related to arteri- and coronaviruses
More LessA 20089 nucleotide (nt) sequence was determined for the 5′ end of the (+)-ssRNA genome of gill-associated virus (GAV), a yellow head-like virus infecting Penaeus monodon prawns. Clones were generated from a ∼22 kb dsRNA purified from lymphoid organ total RNA of GAV-infected prawns. The region contains a single gene comprising two long overlapping open reading frames, ORF1a and ORF1b, of 4060 and 2646 amino acids, respectively. The ORFs are structurally related to the ORF1a and ORF1ab polyproteins of coronaviruses and arteriviruses. The 99 nt overlap between ORF1a and ORF1b contains a putative AAAUUUU ‘slippery’ sequence associated with −1 ribosomal frameshifting. A 131 nt stem–loop with the potential to form a complex pseudoknot resides 3 nt downstream of this sequence. Although different to the G/UUUAAAC frameshift sites and ‘H-type’ pseudoknots of nidoviruses, in vitro transcription/translation analysis demonstrated that the GAV element also facilitates read-through of the ORF1a/1b junction. As in coronaviruses, GAV ORF1a encodes a 3C-like cysteine protease domain located between two hydrophobic regions. However, its sequence suggests some structural relationship to the chymotrypsin-like serine proteases of arteriviruses. ORF1b encodes homologues of the ‘SDD’ polymerase, which among (+)-RNA viruses is unique to nidoviruses, as well as metal-ion-binding and helicase domains. The presence of a dsRNA replicative intermediate and ORF1a and ORF1ab polyproteins translated by a −1 frameshift suggests that GAV represents the first invertebrate member of the Order Nidovirales.
-
-
-
Leader switching occurs during the rescue of defective RNAs by heterologous strains of the coronavirus infectious bronchitis virus
More LessA defective RNA (D-RNA), CD-61, derived from the Beaudette strain of the avian coronavirus infectious bronchitis virus (IBV), was rescued (replicated and packaged) using four heterologous strains of IBV as helper virus. Sequence analysis of the genomic RNA from the four heterologous IBV strains (M41, H120, HV10 and D207) identified nucleotide differences of up to 17% within the leader sequence and up to 4·3% within the whole of the adjacent 5′ untranslated region (UTR). Analysis of the 5′ ends of the rescued D-RNAs showed that the Beaudette leader sequence, present on the initial CD-61, had been replaced with the corresponding leader sequence from the helper IBV strain but the adjacent 5′ UTR sequence of the rescued D-RNAs corresponded to the original CD-61 Beaudette sequence. These results demonstrated that the phenomenon of leader switching previously identified for the coronaviruses murine hepatitis virus and bovine coronavirus (BCoV) also occurred during the replication of IBV D-RNAs. Three predicted stem–loop structures were identified within the 5′ UTR of IBV. Stem–loop I showed a high degree of covariance amongst the IBV strains providing phylogenetic evidence that this structure exists and is potentially involved in replication, supporting previous observations that a BCoV stem–loop homologue was essential for replication of BCoV defective interfering RNAs.
-
-
-
Characterization of the sialic acid binding activity of transmissible gastroenteritis coronavirus by analysis of haemagglutination-deficient mutants
More LessTransmissible gastroenteritis coronavirus (TGEV) agglutinates erythrocytes of several species by virtue of sialic acid binding activity of the surface protein S. We have isolated and characterized five haemagglutination-defective (HAD) mutants. In contrast to the parental virus, the mutants were unable to bind to porcine submandibulary mucin, a substrate rich in sialic acid. Each of the mutants was found to contain a single point mutation in the S protein (Cys155Phe, Met195Val, Arg196Ser, Asp208Asn or Leu209Pro), indicating that these amino acids are affecting the sialic acid binding site. In four of the HAD mutants a nearby antigenic site is affected in addition to the sialic acid binding site, as indicated by reactivity with monoclonal antibodies. The parental virus was found to have an increased resistance to the detergent octylglucoside compared to the HAD mutants. This effect depended on cellular sialoglycoconjugates bound to the virion. If the binding of sialylated macromolecules was prevented by neuraminidase treatment, the parental virus was as sensitive to octylglucoside as were the HAD mutants. We discuss the possibility that the sialic acid binding activity helps TGEV to resist detergent-like substances encountered during the gastrointestinal passage and thus facilitates the infection of the intestinal epithelium. An alternative function of the sialic acid binding activity – accessory binding to intestinal tissues – is also discussed.
-
-
-
Clearance of infection in cats naturally infected with feline coronaviruses is associated with an anti-S glycoprotein antibody response
More LessWe have investigated by Western blotting the antibody responses against the three major structural proteins in cats naturally infected with feline coronaviruses that cleared virus infection (group I), established chronic asymptomatic infection (group II) or were sick (group III). The cats of group I developed an anti-S glycoprotein response that was, relative to the anti-M glycoprotein response, at least 30-fold higher than that of chronically infected cats from groups II and III. These results suggest that the anti-S glycoprotein response against antigenic domains revealed by Western blot is associated with clearance of the virus after natural infection, and is not a risk factor for the establishment of a chronic infection.
-
-
-
Identification and subcellular localization of a 41 kDa, polyprotein 1ab processing product in human coronavirus 229E-infected cells.
More LessThe translation products of the human coronavirus (HCV) 229E open reading frames 1a and 1b, the polyproteins 1a and 1ab, are processed by virus- encoded proteinases. One of the key enzymes in this process is a chymotrypsin-like enzyme, the 3C- like proteinase. In this study we have identified an ORF 1b-encoded, 41 kDa processing product in HCV 229E-infected cells by using a monoclonal antibody with defined specificity. We show that this polypeptide is released from polyprotein 1ab by 3C-like proteinase-mediated cleavage of the peptide bonds Gln-6110/Gly-6111 and Gln-6458/Ser- 6459. Also, we have investigated the subcellular localization of the 41 kDa processing product. Immunofluorescence microscopy revealed a punctate, perinuclear distribution of the 41 kDa polypeptide in infected cells and an identical subcellular localization was observed for three additional pp1ab-derived polypeptides. In contrast, the virus nucleocapsid protein showed a homogeneous cytoplasmic localization.
-
-
-
Identification of residues critical for the human coronavirus 229E receptor function of human aminopeptidase N.
More LessAminopeptidase N (APN) is the major cell surface receptor for group 1 coronaviruses. In this study, we have isolated and characterized a feline APN cDNA and shown that the transfection of human embryonic kidney cells with this cDNA renders them susceptible to infection with the feline coronavirus feline infectious peritonitis virus, the human coronavirus (HCV) 229E and the porcine coronavirus porcine transmissible gastroenteritis virus. By using chimeric APN genes, assembled from porcine and feline sequences, we have shown that, analogously to the human APN protein, a region within the amino-terminal part of the feline APN protein (encompassing amino acids 132–295) is essential for its HCV 229E receptor function. Furthermore, by comparing the relevant feline, human and porcine APN sequences, we were able to identify a hypervariable stretch of eight amino acids that are more closely related in the feline and human APN proteins than in the porcine APN molecule. Using PCR- directed mutagenesis, we converted this stretch of amino acids within the porcine APN molecule to the corresponding residues of the human APN molecule. These changes were sufficient to convert porcine APN into a functional receptor for HCV 229E and thus define a small number of residues that are critically important for the HCV 229E receptor function of human APN.
-
-
-
No evidence for quasispecies populations during persistence of the coronavirus mouse hepatitis virus JHM: sequence conservation within the surface glycoprotein gene S in Lewis rats
More LessThe surface glycoprotein S (spike) of coronaviruses is believed to be an important determinant of virulence and displays extensive genetic polymorphism in cell culture isolates. This led us to consider whether the observed heterogeneity is reflected by a quasispecies distribution of mutated RNA molecules within the infected organ. Corona- virus infection of rodents is a useful model system for investigating the pathogenesis of virus-induced central nervous system (CNS) disease. Here, we investigated whether genetic changes in the S gene occurred during virus persistence in vivo. We analysed the variability of S gene sequences directly from the brain tissue of Lewis rats infected with the coronavirus mouse hepatitis virus (MHV) variant JHM-Pi using RT-PCR amplification methods. The S gene sequence displayed a remarkable genetic stability in vivo. No evidence for a quasispecies distribution was found by sequence analysis of amplified S gene fragments derived from the CNS of Lewis rats. Furthermore, the S gene also remained conserved under the selection pressure of a neutralizing antibody. Only a few mutations predicted to result in amino acid changes were detected in single clones. The changes were not represented in the consensus sequence. These results indicate that to retain functional proteins under the constraints of a persistent infection in vivo, conservation of sequence can be more important than heterogeneity.
-
-
-
Expression and characterization of a recombinant murine coronavirus 3C-like proteinase
More LessThe replication of coronaviruses involves proteolytic processing of the gene 1 translation products, pp1a and pp1ab. One of the key enzymes in this process is predicted to be a virus-encoded 3C-like proteinase. In this report, we describe a bacterial system that has allowed us to express and characterize a recombinant murine coronavirus (MHV- JHM) 3C-like proteinase. The partially purified protein has been shown to exhibit proteolytic activity in trans and mutation analysis has been used to demonstrate the indispensability of Cys- 3495 for enzymatic activity. Finally, the effect of class-specific proteinase inhibitors on the trans cleavage activity of the MHV 3C-like proteinase has been used to demonstrate the functional and structural homology of this enzyme to the picorna- virus 3C proteinases.
-
-
-
Characterization of functional domains in the human coronavirus HCV 229E receptor
More LessHuman aminopeptidase N (hAPN or CD13) and porcine aminopeptidase N (pAPN) are functional receptors for human coronavirus (HCV) 229E and porcine transmissible gastroenteritis virus (TGEV), respectively. However, hAPN cannot function as a receptor for TGEV and pAPN cannot function as a receptor for HCV 229E. In this study, we constructed a series of chimeric hAPN/pAPN genes and expressed the corresponding proteins in transfected cells. Subsequently, we identified the chimeric proteins that can function as a receptor for HCV 229E. The results show that replacement of a small region of pAPN sequence (pAPN amino acids 255–348) with the corresponding hAPN sequence (hAPN amino acids 260–353) converts pAPN into a functional receptor for HCV 229E. The region of hAPN that we have defined in this way does not correspond to the region of pAPN that has been identified as essential for the TGEV-receptor interaction. We conclude that although both viruses use a homologous receptor protein, different regions of the protein are required to mediate susceptibility to infection with HCV 229E and TGEV.
-
-
-
Virus entry into a polarized epithelial cell line (MDCK): similarities and dissimilarities between influenza C virus and bovine coronavirus
More LessWe have analysed the uptake of influenza C virus and bovine coronavirus (BCV) by a polarized epithelial cell line, Madin-Darby canine kidney (MDCK) cells. Both viruses use N-acetyl-9-O-acetyl-neuraminic acid as a receptor determinant for attachment to cells. Virus binding assays with immobilized proteins indicated that a glycoprotein of 40 kDa is the major surface protein containing the receptor determinant for the two viruses. MDCK cells grown on filters for permeable support were found to have differential sensitivity to infection by these viruses. Both viruses were able to initiate infection via the apical domain of the plasma membrane, but only influenza C virus also accomplished infection via the basolateral plasma membrane. The resistance of MDCK cells to BCV infection from the basal filter chamber was overcome when the cell polarity was abolished by maintaining the cells in calcium-free medium. This finding indicates that the resistance to basolateral infection by BCV is a property of the cell line and not due to a technical problem related to the use of filters. Our results indicate that two viruses which use the same receptor for attachment to cells may differ in their ability to enter polarized cells. The possible involvement of an accessory molecule in the entry of BCV is discussed.
-
-
-
The region between the M and S genes of porcine haemagglutinating encephalomyelitis virus is highly similar to human coronavirus OC43
More LessThe nucleotide sequences of the regions between the membrane and spike protein genes of three strains of porcine haemagglutinating encephalomyelitis virus (HEV) were determined. A total of 739 (HEV strain 67N) and 751 (strains NT9 and VW572) nucleotides were sequenced. Two ORFs, potentially encoding proteins of 12.8 and 9.6 kDa, were identified. Pairwise comparisons with the corresponding ORFs in bovine coronavirus (BCV) and human coronavirus (HCV) OC43 revealed sequence similarities of greater than 88.5% at the nucleotide and 85.3% at the amino acid level for the 12.8 kDa ORF product. For the 9.6 kDa ORF product similarities were greater than 96.9% and 95.2%, respectively. An additional 12 nucleotide deletion upstream of the 12.8 kDa ORF start codon was found in HEV 67N compared to NT9 and VW572. These results reveal a genomic organization of HEV in the region analysed that is homologous to HCV OC43 but different from BCV.
-
-
-
An adenovirus recombinant expressing the spike glycoprotein of porcine respiratory coronavirus is immunogenic in swine
More LessThe full-length spike (S) gene of porcine respiratory coronavirus (PRCV) was inserted into the genome of human adenovirus type 5 downstream of the early transcription region 3 promoter. The recombinant virus replicated in cultures of the swine testicle ST cell line and directed the synthesis of S antigen with a maximum yield of approximately 26 µg per 106 cells. The antigen was cell-associated except in the late phase of the infection, when a small amount (3.5 µg per 106 cells) was released. The cell-associated antigen consisted of polypeptides of molecular mass 160 kDa and 175 kDa, comigrating with the authentic precursor S′ and the mature S protein of PRCV, respectively. The extracellular recombinant antigen corresponded to the 175 kDa mature protein. Some recombinant S protein was exposed on the cell surface and was recognized by neutralization-mediating anti-S monoclonal antibodies. Piglets, inoculated oronasally with the recombinant adenovirus vector developed PRCV-neutralizing serum antibodies and were partially protected against PRCV challenge, demonstrating the potential of live adenovirus as vaccine vector.
-
-
-
A region of the coronavirus infectious bronchitis virus 1a polyprotein encoding the 3C-like protease domain is subject to rapid turnover when expressed in rabbit reticulocyte lysate
More LessIn order to investigate the mechanisms involved in the processing of infectious bronchitis virus polyproteins, several candidate regions of the genome have been cloned and expressed in vitro. During these studies it was observed that the translation product encoded by one of these clones (pKT205) was poorly expressed. Biochemical and genetic analyses revealed that the basis for the poor expression was a post-translational event involving ubiquitination of the protein and degradation by an ATP-dependent system operating in the reticulocyte lysate used for the in vitro expression. Two independently acting regions which conferred instability were identified, one of which mapped to the predicted 3C protease domain, contained within the 5′ end of the clone, while the other, more C-terminal region, was effective in conferring instability upon a heterologous protein to which it had been transferred. These regions may influence the stability of the authentic viral protein(s) in vivo and hence allow for the control of their expression and/or function at the level of proteolysis by cellular protease(s).
-
-
-
Comparison of bovine coronavirus isolates associated with neonatal calf diarrhoea and winter dysentery in adult dairy cattle in Québec
More LessCytopathic coronaviruses were isolated in HRT-18 cells from bloody faecal samples collected from cows in Québec dairy herds having experienced typical outbreaks of winter dysentery (WD). The formation of polykaryons in the infected cell cultures was found to be dependent on the presence of trypsin in the medium. The WD isolates differed from the prototype Mebus strain of bovine enteropathogenic coronavirus (BCV.Meb) in respect to haemagglutination inhibition (HI), haemagglutination patterns at 4 °C and 37 °C, and receptor destroying enzyme activity with rat erythrocytes. Other field strains of BCV associated with outbreaks of neonatal calf diarrhoea (NCD) also differed from the BCV.Meb strain by demonstrating differences in HI. In all cases, no differences were detected by virus neutralization and Western immunoblotting. Analysis and comparison of the nucleotide and deduced amino acid sequences of the PCR-amplified haemagglutinin esterase (HE) genes of one representative WD strain (BCQ.2590) and two highly cytopathic NCD strains (BCQ.3 and BCQ.571) revealed high degrees of similarities (nt and aa sequence homologies > 98%) with the BCV.Meb strain. The putative esterase active site FGDS was conserved among these four BCV strains, indicating that this domain is probably not a determinant for BCV virulence. Six amino acid substitutions occurred between the HE glycoproteins of BCV.Meb and BCQ.2590 strains; two proline substitutions occurred respectively in the signal peptide (at aa 5) and near the sequences of the putative esterase domain (at aa 53).
-
-
-
Sequence and expression of the ns2 protein gene of human coronavirus OC43
More LessThe complete nucleotide sequence of the ns2 gene of human coronavirus OC43 (HCV-OC43) was determined. Sequence analysis revealed an open reading frame that could encode a protein of 278 amino acids, with an estimated molecular mass of 32.2 kDa. Six potential phosphorylation sites are present but no sites of N-glycosylation were found. The amino acid sequence of the HCV-OC43 ns2 protein shows 92% identity with that of the Mebus strain of bovine coronavirus (BCV). However, a stretch of nine consecutive amino acids near the C terminus is completely different, causing it to be very hydrophilic, which contrasts with the hydrophobic nature of this region in BCV. As shown by immunofluorescence with a monospecific antiserum, the ns2 protein was expressed in the cytoplasm of HCV-OC43-infected HRT-18 cells.
-
