Coronaviruses
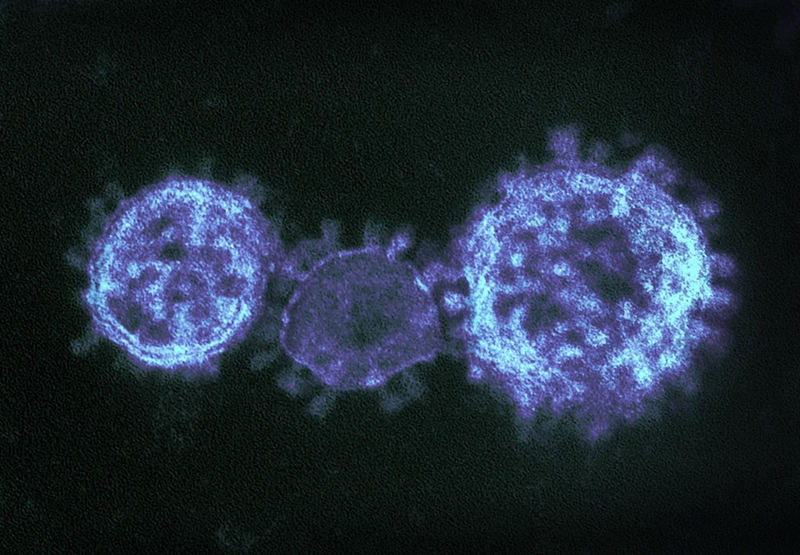
Coronaviruses are a large family of viruses that can infect a range of hosts. They are known to cause diseases including the common cold, Severe Acute Respiratory Syndrome (SARS) and Middle East Respiratory Syndrome (MERS) in humans.
In January 2020, China saw an outbreak of a new coronavirus strain now named SARS-CoV-2. Although the animal reservoir for the SARS and MERS viruses are known, this has yet to have been confirmed for SARS-CoV-2. All three strains are transmissible between humans.
To allow the widest possible distribution of relevant research, the Microbiology Society has brought together articles from across our portfolio and made this content freely available.
Image credit: "MERS-CoV" by NIAID is licensed under CC BY 2.0, this image has been modified.
Collection Contents
201 - 250 of 298 results
-
-
Characterization of the IgA and subclass IgG responses to neutralizing epitopes after infection of pregnant sows with the transmissible gastroenteritis virus or the antigenically related porcine respiratory coronavirus
More LessIn this study, we have investigated the characteristics of secreted IgA and other classes of Ig induced after vaccination of sows with transmissible gastroenteritis virus (TGEV) or the antigenically related porcine respiratory coronavirus (PRCV). Both viruses induced the secretion of neutralizing antibodies of different classes in the sows’ milk, but these protected suckling piglets against TGEV to different degrees. Quantitative differences in the induction of IgA by both viruses were found among the different viral antigenic sites and subsites of glycoprotein S. In TGEV-vaccinated sows, antigenic subsite A was the best inducer of IgA, followed by antigenic site D. After vaccination with PRCV, lower levels of IgA were detected on colostrum and milk, antigenic site D and subsite Ab being the immunodominant sites. This quantitative difference in epitope recognition could explain the differences in newborn piglet protection found using Ig classes purified from the milk of sows immunized with both viruses. Apparently only IgA recognizing at least antigenic sites A and D confers good protection in vivo, whereas any Ig class recognizing only one antigenic site may neutralize the virus in cell culture. These results indicate that the formulation of a subunit vaccine against TGEV has to consider the inclusion of more than one antigenic site involved in virus neutralization.
-
-
-
Nucleotide sequence and expression of the spike (S) gene of canine coronavirus and comparison with the S proteins of feline and porcine coronaviruses
More LessWe have cloned, sequenced and expressed the spike (S) gene of canine coronavirus (CCV; strain K378). Its deduced amino acid sequence has revealed features in common with other coronavirus S proteins: a stretch of hydrophobic amino acids at the amino terminus (the putative signal sequence), another hydrophobic region at the carboxy terminus (the membrane anchor), heptad repeats preceding the anchor, and a cysteine-rich region located just downstream from it. Like other representatives of the same antigenic cluster (CCV-Insavc-1 strain, feline infectious peritonitis and enteric corona- viruses, porcine transmissible gastroenteritis and respiratory coronaviruses, and the human coronavirus HCV 229E), the CCV S polypeptide lacks a proteolytic cleavage site present in many other coronavirus S proteins. Pairwise comparisons of the S amino acid sequences within the antigenic cluster demonstrated that the two CCV strains (K378 and Insavc-1) are 93·3% identical, about as similar to each other as they are to the two feline coronaviruses. The porcine sequences are clearly more divergent mainly due to the large differences in the amino-terminal (residues 1 to 300) domains of the proteins; when only the carboxy-terminal parts (residues 301 and on) are considered the homologies between the canine, feline and porcine S polypeptides are generally quite high, with identities ranging from 90·8 % to 96·8 %. The human coronavirus is less related to the other members of the antigenic group. A phylogenetic tree constructed on the basis of the S sequences showed that the two CCVs are evolutionarily more related to the feline than to the porcine viruses. Expression of the CCV S gene using the vaccinia virus T7 RNA polymerase system yielded a protein of the expected M r (approximately 200K) which could be immunoprecipitated with an anti-feline infectious peritonitis virus polyclonal serum and which was indistinguishable from the S protein synthesized in CCV-infected cells.
The nucleotide sequence data presented in this paper have been submitted to the EMBL database and assigned the accession number X77047.
-
-
-
Expression of the S1 and S2 subunits of murine coronavirus JHMV spike protein by a vaccinia virus transient expression system
More LessThe spike (S) protein of murine coronavirus JHMV, variant cl-2, comprises two polypeptides, N-terminal S1 (with an N-terminal signal peptide) and C-terminal S2 (with a C-terminal transmembrane domain). In order to express these subunits, we constructed three different vaccinia virus transfer vectors (VV-TVs) containing cDNAs encoding the S1 protein without a transmembrane domain (pSFS1utt), the S1 protein with a C-terminal transmembrane domain derived from S2 (pSFS1tmd) or the S2 protein with an N-terminal signal peptide derived from S1 (pSFssS2). The S1 and S2 proteins were expressed in DBT cells by infection with vaccinia virus and transfection of these VV-TVs. In cells transfected with the pSFS1utt and pSFS1tmd, 96K and 106K proteins, respectively, were detected by Western blotting. The ssS2 protein expressed by pSFssS2 was 96K, which was slightly larger than the authentic S2 protein. The S1utt and S1tmd proteins were shown by binding studies using a panel of monoclonal antibodies to be antigenically indistinguishable from the authentic S1 protein. The S1tmd and ssS2 proteins were detected on the cell surface by immunofluorescence, whereas the S1utt protein was not. However, when the S1utt protein was expressed together with the ssS2 protein, the S1utt was detected on the cell membrane. This suggested that the S1utt was associated with ssS2 on the cell membrane. These observations indicate that the expressed S1 and S2 proteins associated in a similar manner to the authentic S1 and S2 proteins produced in DBT cells infected with cl-2. However, cell fusion was not observed in cells expressing either S1 or S2 nor in cells coexpressing both S1 and S2, although the whole S protein expressed by VV-TV did induce fusion.
-
-
-
Molecular characterization of the S protein gene of human coronavirus OC43
More LessThe gene encoding the spike protein of the OC43 strain of human coronavirus (HCV-OC43) was cloned and sequenced. The complete nucleotide sequence revealed an open reading frame of 4062 nucleotides encoding a protein of 1353 amino acids with a predicted M r of 150078. Structural features include 22 N-glycosylation sites, an N-terminal hydrophobic signal sequence of 17 amino acids, an hydrophilic cysteine-rich sequence of 35 amino acids near the C terminus, and a potential proteolytic cleavage site (RRSR) between amino acid residues 758 and 759, yielding S1 and S2 segments of 84730 and 65366 M r, respectively. The predicted amino acid sequence of the spike protein of HCV-OC43 has 91% identity with that of the Mebus strain of bovine coronavirus, revealing more sequence divergence in the putative bulbous part (S1) than in the predicted stem region (S2).
-
-
-
Sequence determination of the nucleocapsid protein gene of the porcine epidemic diarrhoea virus confirms that this virus is a coronavirus related to human coronavirus 229E and porcine transmissible gastroenteritis virus
More LessThe nucleotide sequence of 1.7 kbp cDNA, comprising the region nearest the 3′ end of the genome of the porcine epidemic diarrhoea virus (PEDV), has been independently determined for two European isolates of PEDV. Almost identical results were obtained for the two isolates, which were derived from cases of PEDV infection in Belgium and Britain in 1977 and 1987, respectively. The sequences contained a 1323 nucleotide (nt) open reading frame (ORF), which showed moderate identity to the nucleocapsid (N) gene of other coronaviruses. The greatest similarity at both the nucleic acid and protein levels was to the human coronavirus 229E. The PEDV N gene was, however, notably larger than that of the human 229E and porcine transmissible gastroenteritis viruses. This reflects the presence of a putative insertion of approximately 135 nt located towards the middle of the N gene. A second 336 nt ORF, which might encode a leucine-rich protein similar to, but shorter than, the bovine coronavirus internal protein was found within the PEDV N gene. Several RNA motifs typical of coronaviruses were also observed. These results confirm the earlier provisional classification of PEDV as a coronavirus.
-
-
-
An immunodominant CD4+ T cell site on the nucleocapsid protein of murine coronavirus contributes to protection against encephalomyelitis
More LessThe murine coronavirus neurotropic strain JHM (MHV-JHM) nucleocapsid (N) protein induces a strong T-helper cell response in Lewis rats. It has been shown previously that N-specific CD4+ T cells can confer protection against acute disease upon transfer to otherwise lethally infected rats. To define the major antigenic regions that elicit this T cell response, truncated fragments of N protein were expressed from a bacterial expression vector and employed as T cell antigens. Lymphocytes from either MHV-JHM-infected or immunized rats were stimulated in culture with virus antigen, grown and tested for their specificity to the N protein fragments. The carboxy-terminally located C4-N fragment (95 amino acids) induced the most pronounced proliferative response irrespective of whether the lymphocyte culture was derived from immunized or MHV-JHM-infected rats. We established T cell lines specific for the truncated N protein fragments and tested their potential to mediate protection by transfer experiments. Only the T cell line C4-N and the T cell line specific for the full-length N protein were protective. By contrast, all truncated N protein fragments elicited a humoral immune response and contained antigenic sites recognized by antibodies from diseased rats.
-
-
-
Neutralization and fusion inhibition activities of monoclonal antibodies specific for the S1 subunit of the spike protein of neurovirulent murine coronavirus JHMV c1-2 variant
More LessThe cleavage products of the spike (S) protein, the S1 and S2 subunits, of the highly neurovirulent murine coronavirus (MHV) JHMV c1-2 variant were identified by immunoprecipitation of virus-infected cell lysates after treatment with urea and 2-mercaptoethanol. By this method 14 monoclonal antibodies (MAbs) raised against the S protein of the c1-2 variant were revealed to react with the S1 subunit and one with the S2 subunit. These 14 MAbs were classified into the following three groups: (A) MAbs reactive to almost all MHV strains examined, (B) MAbs specific for the JHMV strain and (C) MAbs specific for a large S protein of the JHMV strain. All five MAbs classified in group B showed neutralization activity and four of them also showed fusion inhibition activity. Four of six MAbs in group C showed neutralizing activity to the c1-2 variant but not to the sp-4 variant, and most of them had no fusion inhibition activity. Western blot analyses showed that all of the MAbs, except for no. 2 in group A, failed to react with the denatured S and S1 proteins. All MAbs in groups A and C, with the exception of no. 19 in group A, reacted with the mildly denatured S proteins, whereas none of the MAbs in group B did. These results suggest that MAbs in group B recognized highly conformational epitopes which may be involved in the binding of virions to cellular receptors and the fusion activity of the virus.
-
-
-
Proteolytic cleavage of the murine coronavirus surface glycoprotein is not required for fusion activity
More LessA cDNA copy of the murine coronavirus [otherwise known as murine hepatitis virus (MHV)] surface (S) glycoprotein gene was isolated and expressed in DBT cells by using a recombinant vaccinia virus system. The expressed S protein induced extensive syncytium formation at neutral pH. Oligonucleotide mutagenesis was used to engineer an S protein gene in which codons for the proteolytic cleavage site, Arg-Arg-Ala-Arg-Arg, were replaced with an equal number of codons for amino acids with aliphatic or aliphatic hydroxyl side-chains. The mutated S protein was stably expressed in DBT cells and, in contrast to the wild-type protein, was not proteolytically cleaved. Nevertheless, the non-cleaved protein induced extensive syncytium formation. These results clearly indicate that the non-cleaved form of the MHV S protein is able to mediate cell membrane fusion. Thus proteolytic cleavage is not an absolute requirement for fusion activity.
-
-
-
Bovine coronavirus spike glycoprotein: localization of an immunodominant region at the amino-terminal end of S2
More LessWe have identified the binding site of monoclonal antibodies (MAbs) against the S2 subunit of the bovine coronavirus spike (S) glycoprotein. The location of this site was first investigated by using prokaryotic expression of DNA restriction fragments covering the entire S gene. The amino acid sequence containing the antibody binding site was shortened from 70 to 20 amino acids by digestion of plasmid DNA with exonuclease III, followed by sequencing of the smallest digestion product encoding an immunoreactive fusion protein. Finally we synthesized a set of nonapeptides covering the 20 amino acid sequence extending from the N-terminal residue of the S2 subunit (Ala 769 to Tyr 798). MAbs reacted mainly with six consecutive overlapping peptides with the sequence TTGYRFTN-FEPFTV. Polyclonal antibodies from hyperimmunized or convalescent animals reacted only with the recombinant proteins identified by MAbs, and the hyperimmune serum bound to the same set of peptides. This suggests that this highly conserved linear antigenic determinant corresponds to an immunodominant region. This region resembles both in location and immunodominance the linear determinant defined on the infectious bronchitis virus S2 subunit. The presence of similar regions in the N-terminal region of the S2 subunit of other coronaviruses is discussed.
-
-
-
Differential in vitro inhibition of feline enteric coronavirus and feline infectious peritonitis virus by actinomycin D
More LessThe growth of feline enteric coronavirus strain 79-1683 in whole feline embryo cells was inhibited by the presence of 1 µg/ml of actinomycin D in the culture fluid. No virus-specific mRNAs could be detected in such cultures and yields of infectious virus were depressed by >99%. By contrast, the antigenically related feline infectious peritonitis virus strain 79-1146 was unaffected by the presence of actinomycin D, indicating a fundamental difference between the two feline coronavirus strains in their requirements for host-encoded function(s).
-
-
-
Analysis of a 9.6 kb sequence from the 3′ end of canine coronavirus genomic RNA
More LessWe have analysed the organization of the 3′ end of the genomic RNA of canine coronavirus (CCV), a virus which has a close antigenic relationship to transmissible gastroenteritis virus (TGEV), porcine respiratory coronavirus (PRCV) and feline infectious peritonitis virus (FIPV). Genomic RNA isolated from CCV strain Insavc-1-infected A72 cells was used to generate a cDNA library. Overlapping clones, spanning approximately 9.6 kb [from the 3′ end of the polymerase gene, 1b, to the poly(A) tail] were identified. Sequencing and subsequent analyses revealed 10 open reading frames (ORFs). Three of these code for the major coronavirus structural polypeptides S, M and N; a fourth codes for a small membrane protein, SM, a putative homologue of the IBV structural polypeptide 3c, and five code for polypeptides, designated 1b, 3a, 4, 7a and 7b, homologous to putative non-structural polypeptides encoded in the TGEV or FIPV genomes. An extra ORF which had not hitherto been identified in this antigenic group of coronaviruses was designated 3x. Pairwise alignment of these ORFs with their counterparts in TGEV, PRCV and FIPV revealed high levels of identity and highlighted the close relationship between the members of this group of viruses.
-
-
-
Sequence analysis of the membrane protein gene of human coronavirus OC43 and evidence for O-glycosylation
More LessThe gene encoding the membrane (M) protein of the OC43 strain of human coronavirus (HCV-OC43) was amplified by a reverse transcription-polymerase chain reaction of viral RNA with HCV-OC43- and bovine coronavirus (BCV)-specific primers. The nucleotide sequence of the cloned 1.5 kb fragment revealed an open reading frame (ORF) of 690 nucleotides which was identified as the M protein gene from its homology to BCV. This ORF encodes a protein of 230 amino acids with an M r of 26416. The gene is preceded by the motif UCCAAAC, analogous to the consensus coronavirus transcription initiation sequence. The M protein of HCV-OC43 shows features typical of all coronavirus M proteins studied: a hydrophilic, presumably external N terminus including about 10% of the protein, and a potential N-glycosylation site followed by three major hydrophobic transmembrane domains. The amino acid sequence of the M protein of HCV-OC43 has 94% identity with that of the Mebus strain of BCV, and also contains six potential O-glycosylation sites in the exposed N-terminal domain. Indeed, the glycosylation of the M protein was not inhibited in the presence of tunicamycin, which is indicative of O-glycosylation, as previously reported for BCV and murine hepatitis virus. Virions released from tunicamycin-treated cells contained the M glycoprotein but were devoid of both peplomer (S) and haemagglutinin-esterase (HE) proteins. Thus, inhibition of the N-glycosylation of the S and HE structural proteins prevented their incorporation into progeny virions, an indication that they are dispensable for virion morphogenesis, unlike the M protein.
-
-
-
Synthesis and Processing of the Haemagglutinin—esterase Glycoprotein of Bovine Coronavirus Encoded in the E3 Region of Adenovirus
More LessThe haemagglutinin—esterase gene (HE) of bovine coronavirus (BCV) encodes a major viral membrane glycoprotein that elicits BCV-neutralizing antibodies. The BCV HE gene was cloned into a human adenovirus serotype 5 (Ad5) transfer vector in place of early transcription region 3, and a helper-independent recombinant virus was constructed by rescue of the transcription unit by homologous in vivo recombination between the vector and Ad5 genomic DNA. The BCV HE polypeptide expressed by this recombinant Ad was characterized in vivo and in vitro. A 65K polypeptide was identified using an anti-BCV antibody in both human (293) and bovine (MDBK) cells infected with the recombinant Ad. In the absence of a reducing agent, migration of the 65K polypeptide was shifted to 130K, indicating that the recombinant HE polypeptide existed in a dimeric form. The HE polypeptide was glycosylated, as demonstrated by labelling with [3H]glucosamine, and was immunoreactive with three distinct groups of conformation-specific anti-HE monoclonal antibodies (MAbs). Cells infected with recombinant Ad expressing BCV HE exhibited both haemadsorption activity and acetylesterase activity. In addition, the anti-HE group A MAbs HC10-5 and KD9-40 inhibited both the haemadsorption activity and esterase activity of the recombinant HE polypeptide, suggesting that the antigenic domain responsible for BCV neutralization may overlap (or is closely associated with) the domain(s) responsible for haemagglutination and/or acetylesterase activities. When mice were inoculated intraperitoneally with live recombinant Ad, a significant level of BCV-neutralizing HE-specific antibody was induced. These results indicate that the recombinant Ad replicates and directs the synthesis of the BCV HE polypeptide in vivo.
-
-
-
Bovine coronavirus peplomer glycoproteins: detailed antigenic analyses of S1, S2 and HE
More LessForty-four monoclonal antibodies (MAbs) to the G110 isolate of bovine enteric coronavirus were used for the characterization of the peplomer proteins S and HE. Fourteen of these MAbs reacted with HE and the remaining 30 with the products of the S gene, S1 (19 MAbs), S2 (six MAbs) and gp200 (five MAbs). S1 and HE were found to carry major neutralization determinants, and S1 appeared to elicit the production of the MAbs displaying the highest neutralizing activity. The topography of the epitopes was assessed by means of a competitive binding assay; the 44 MAbs defined four independent antigenic domains on S1, two on S2, one on gp200 and two on HE. All the neutralizing anti-S1 MAbs mapped in antigenic sites A and B and all the neutralizing anti-HE MAbs in HE-B. Antigenic site S1-B was further subdivided into four subsites. Functional mapping was performed by testing a library of neutralization-resistant mutants against the neutralizing MAbs. Analysis of their reactivity in a neutralization test confirmed the overall distribution of epitopes in S1-B and HE-B.
-
-
-
Molecular cloning and expression of a spike protein of neurovirulent murine coronavirus JHMV variant c1-2
More LessA cDNA encoding the spike (S) protein of the neurovirulent murine coronavirus JHMV variant c1-2 was isolated and sequenced. Analysis of the cDNA revealed that the S protein consists of 1376 amino acids, as does the S protein of mouse hepatitis virus 4. We inserted the cDNA into the genome of vaccinia virus to obtain a recombinant vaccinia virus (rVV). The S protein expressed in RK13 cells infected by the rVV was shown to be electrophoretically and immunologically indistinguishable from the S protein produced in DBT cells infected with c1-2 virus. RVV infection of rats and mice induced S protein-specific antibody production detectable by immunofluorescence and neutralization. Moreover, the S protein expressed by the rVV induced syncytium formation not only in mouse DBT and L cells, which are susceptible to c1-2 virus infection, but also in rabbit RK13 cells, which are not susceptible to c1-2 virus infection. This result suggests the possibility that RK13 cells have binding sites for the c1-2 virus S protein.
-
-
-
Bovine coronavirus uses N-acetyl-9-O-acetylneuraminic acid as a receptor determinant to initiate the infection of cultured cells
More LessThe importance of N-acetyl-9-O-acetylneuraminic acid (Neu5,9Ac2) as a receptor determinant for bovine coronavirus (BCV) on cultured cells was analysed. Pretreatment of MDCK I (Madin Darby canine kidney) cells with neuraminidase or acetylesterase rendered the cells resistant to infection by BCV. The receptors on a human (CaCo-2) and a porcine (LLC-PK1) epithelial cell line were also found to be sensitive to neuraminidase treatment. The susceptibility to infection by BCV was restored after resialylation of asialo-MDCK I cells with Neu5,9Ac2. Transfer of sialic acid lacking a 9-O-acetyl group was ineffective in this respect. These results demonstrate that 9-O-acetylated sialic acid is used as a receptor determinant by BCV to infect cultured cells. The possibility is discussed that the initiation of a BCV infection involves the recognition of different types of receptors, a first receptor for primary attachment and a second receptor to mediate the fusion between the viral envelope and the cellular membrane.
-
-
-
Monoclonal antibodies differentiate between the haemagglutinating and the receptor-destroying activities of bovine coronavirus
More LessA relatively simple and sensitive method is described which enables the effect of monoclonal antibodies (MAbs) on the receptor-destroying enzyme (RDE) and the haemagglutination (HA) activity of bovine coronavirus (BCV) to be analysed in one assay. A lysate of HRT-18 cells infected with the L9 strain of BCV was found to have a higher RDE:HA ratio than purified virus. At 4 °C the lysate induced an HA pattern which completely disappeared upon raising of the temperature to 37 °C. This L9-infected cell lysate was used to determine the HA inhibition (HAI) titres of MAbs directed against the surface glycoproteins S and HE of BCV. Thereafter, the test plates were incubated at 37 °C to enable the ability of the MAbs to prevent elution of virus from BCV-erythrocyte complexes to be assessed. No inhibition of RDE was detectable with MAbs against glycoprotein S, which had HAI titres ranging from 1:16 to 1:128. On the other hand, MAbs directed against glycoprotein HE had similar HAI titres, but they inhibited elution of 8 HA units of BCV at titres of up to 1:65000.
-
-
-
Sequence analysis of the turkey enteric coronavirus nucleocapsid and membrane protein genes: a close genomic relationship with bovine coronavirus
More LessThe 3′ end of the turkey coronavirus (TCV) genome and the gene encoding the nucleocapsid protein (N) were cloned and sequenced. The gene encoding the membrane protein (M) was obtained by cloning a polymerase chain reaction (PCR)-amplified fragment obtained using bovine coronavirus (BCV)-specific primers. Furthermore, five TCV DNA fragments, obtained by PCR on RNA from clinical specimens and corresponding to either the N terminus of the M protein or the complete M protein were also cloned and sequenced. The sequence revealed a 3′ non-coding region of 291 bases, an open reading frame (ORF) encoding the N protein with a predicted size of 448 amino acids, or an M r of 49K, and an ORF encoding the M protein with a predicted size of 230 amino acids and an M r of 26K. A third ORF, encoding a hypothetical protein of 207 amino acids with an M r of 23K was found within the N gene sequence. The amino acid sequences of both the N and M proteins were more than 99% similar to those published for BCV. Extensive similarity was also observed between the amino acid sequences of the TCV N protein and those of murine hepatitis virus (MHV) (70%) and human respiratory coronavirus strain OC43 (HCV-OC43) (98%) and between the amino acid sequences of the predicted M proteins of TCV and MHV (86%). Such striking identity suggests that BCV, TCV and HCV-OC43 must have diverged from each other only recently. A potential N-glycosylation site was found at the N terminus of the TCV M protein and is situated at the same location in BCV, MHV and transmissible gastroenteritis virus.
-
-
-
High level transient expression of the murine coronavirus haemagglutinin-esterase
More LessWe have expressed the murine coronavirus haemagglutinin-esterase protein in a vaccinia virus/T7 RNA polymerase system. The levels of expression observed are significantly higher than those found in virus-infected cells. The expressed protein has both receptor-destroying (esterase) and receptor-binding (haemad-sorption) activities. The use of this system will greatly facilitate analysis of the structure-function relationships of this protein.
-
-
-
Sequence comparison of the 5′ end of mRNA 3 from transmissible gastroenteritis virus and porcine respiratory coronavirus
More LessAnalysis of porcine transmissible gastroenteritis virus (TGEV) and porcine respiratory coronavirus (PRCV) mRNA species indicated a deletion in mRNA 3 of PRCV. Polymerase chain reaction (PCR) was used to clone the 5′ end of mRNA 3 from PRCV for comparison with the equivalent region in TGEV. Small deletions were observed within and around the PRCV sequence equivalent to the putative open reading frame (ORF) ORF-3a identified in TGEV. The potential RNA polymerase-leader complex binding site (leader RNA binding site), ACTAAAC, found upstream of ORF-3a in TGEV, was absent from the PRCV genome but a potential site was found in the PRCV genome upstream of a gene equivalent to TGEV ORF-3b. PCR analysis, using primers corresponding to sequences within the ORF-3b gene and the leader RNA sequence, confirmed that the leader RNA binding site was upstream of a gene equivalent to TGEV ORF-3b on PRCV mRNA 3 but upstream of ORF-3a on TGEV mRNA 3. The presence of the new leader RNA binding site would be responsible for generating the smaller mRNA 3 species found in PRCV-infected cells.
-
-
-
Porcine respiratory coronavirus differs from transmissible gastroenteritis virus by a few genomic deletions
More LessThe genome organization of porcine respiratory coronavirus (PRCV), a newly recognized agent which has a close antigenic relationship to the enteropathogenic transmissible gastroenteritis virus (TGEV), was studied. Genomic RNA from cell-cultured PRCV (French isolate RM4) was used to produce cDNA clones covering the genomic 3′ end to the start of the spike (S) glycoprotein gene (7519 nucleotides). Six open reading frames (ORFs) were identified that allowed the translation of three coronavirus structural proteins and three putative non-structural (NS) polypeptides, homologous to TGEV ORFs designated NS3-1, NS4 and NS7. Pairwise alignment of PRCV nucleotide and amino acid sequences with sequence data available for three TGEV strains revealed a 96% overall homology. However, the genome of PRCV exhibited two important distinctive features. The first was that the S gene lacked 672 nucleotides in the 5′ region and encoded a truncated form of the S polypeptide, and secondly, the first NS ORF downstream of the S gene was predicted to be non-functional as a consequence of a double deletion. The significance of genomic deletions with respect to tissue tropism and evolution of coronaviruses is discussed.
-
-
-
Four major antigenic sites of the coronavirus transmissible gastroenteritis virus are located on the amino-terminal half of spike glycoprotein S
More LessFour major antigenic sites have been delineated on the spike protein (S) of the porcine enteric coronavirus transmissible gastroenteritis virus (TGEV) in previous topological studies using monoclonal antibodies (MAbs). Correlation of these sites with the physical structure of the protein was achieved by use of different approaches. Recombinant pEX plasmids directing the synthesis of various fused S polypeptides were constructed. A hybrid protein containing nine S-specific residues (363 to 371) was shown to express site C epitopes. The other sites were localized through study of the antigenic activity of fragments generated by controlled cleavage of the native protein with different endopeptidases. Two identified cleavage products of 26K and 13K, immunoreactive to site A-B- and site D-specific MAbs respectively, could be aligned on the S primary structure according to N-terminal sequence data. This led us to propose that the major neutralization domain A-B is contained in a region of approximately 200 residues with residue 506 as its N boundary. Similarly, site D epitopes should be located within a stretch of 130 residues, starting at 82 residues from the N terminus. Point mutations identified by direct RNA sequencing of neutralization-resistant mutants were consistent with the proposed location of these sites.
-
-
-
Nucleotide Sequence of the Gene Encoding the Spike Glycoprotein of Human Coronavirus HCV 229E
More LessThe gene encoding the spike glycoprotein of the human coronavirus HCV 229E has been cloned and sequenced. This analysis predicts an S polypeptide of 1173 amino acids with an Mr of 128600. The polypeptide has 30 potential N -glycosylation sites. A number of structural features typical of coronavirus S proteins can be recognized, including a signal sequence, a membrane anchor, heptad repeat structures and a carboxy-terminal cysteine cluster. A detailed, computer-aided comparison with the S proteins of infectious bronchitis virus, feline infectious peritonitis virus, transmissible gastroenteritis virus and murine hepatitis virus, strain JHM is presented. We have also done a Northern blot analysis of viral RNAs in HCV 229E- infected cells using synthetic oligonucleotides. On the basis of this analysis, and by analogy to the replication strategy of other coronaviruses, we are able to propose a model for the organization and expression of the HCV 229E genome.
-
-
-
Localization of antigenic sites of the E2 glycoprotein of transmissible gastroenteritis coronavirus
More LessFour antigenic sites of the E2 glycoprotein of transmissible gastroenteritis virus were defined by competitive radioimmunoassays of monoclonal antibodies (MAbs). Here, we describe the localization of these sites by testing the antigenicity of protein fragments and prokaryotic expression products of E2 gene fragments, and by sequencing of MAb-resistant (mar) mutants. Partial proteolysis of purified E2 protein allowed the isolation of a 28K fragment recognized by both site A- and site C-specific MAbs. An antiserum against this fragment bound to a synthetic peptide containing residues 1 to 18 and to an expression product containing residues 1 to 325. The same expression product was recognized by site C-specific MAbs. These data indicate that residues within the sequence 1 to 325 contribute to site C and possibly also to site A. Sequencing of mar mutants that escaped neutralization by site A-specific MAbs indicated that residues 538 and 543 also belong to site A. The binding of site-specific MAbs to expression products led directly to the localization of sites B and D, between residues 1 to 325 and 379 to 529, respectively. The first 37 % of the polypeptide chain of E2 appears to be more immunogenic than the rest of the sequence.
-
-
-
Nucleotide sequence of the glycoprotein S gene of bovine enteric coronavirus and comparison with the S proteins of two mouse hepatitis virus strains
More LessThe gene encoding the spike glycoprotein (S) of bovine enteric coronavirus (BECV) was cloned and its complete sequence of 4092 nucleotides was determined. This sequence contained a single long open reading frame with a coding capacity of 1363 amino acids (Mr 150747). The predicted protein had 19 N-glycosylation sites. A signal sequence comprising 17 amino acids was observed starting from the first methionine residue. A potential peptidase cleavage site was located between amino acids 763 and 767. These cleavages explain the maturation of the primary product of the S gene to SI (Mr 104692) and S2 (Mr 84175) spike structural proteins. Two amphipathic α-helices (amino acids 1007 to 1077 and 1269 to 1294) which may constitute the 12 nm stalk of the viral spike were also observed; another a-helix (amino acids 1305 to 1335) may be involved in the anchorage of the spike in the viral membrane. Comparison of this protein sequence to the described homologous mouse hepatitis (MHV) strain A59 and MHV-JHM S protein sequences led us to suggest that MHV-A59 and MHV-JHM S genes could be derived from a deletion of the BECV S gene.
-
-
-
Primary structure of the S peplomer gene of bovine coronavirus and surface expression in insect cells
More LessThe nucleotide sequence of the S peplomer gene of bovine coronavirus (BCV) has been determined. A single open reading frame of 4089 nucleotides encodes a polypeptide of 150K with 20 potential sites for addition of N-linked oligosaccharides. Expression of the cloned BCV S gene by a recombinant of Autographa californica nuclear polyhedrosis virus resulted in production of a 180K glycosylated polypeptide which was transported to the surface of the cell. Comparison of the BCV S gene with the analogous genes of murine hepatitis viruses shows that the BCV S polypeptide contains a unique domain of 138 amino acids not present in murine hepatitis virus strain JHM, but which has a partially homologous counterpart in strain A59. This domain accounts for most of the differences in size of the S gene products of these coronaviruses.
-
-
-
Identification of a New Membrane-associated Polypeptide Specified by the Coronavirus Infectious Bronchitis Virus
More LessNucleotide sequences from the third open reading frame of mRNA D (D3) of infectious bronchitis virus (IBV) were expressed in bacteria as part of a fusion protein with β-galactosidase. Antiserum raised in rabbits against this fusion protein immunoprecipitated from IBV-infected chick kidney or Vero cells a polypeptide of 12·4K, the size expected for a D3- encoded product. The D3 polypeptide is apparently non-glycosylated, and appears to be associated with the membrane fraction of infected cells, as judged by cell fractionation and immunofluorescence.
-
-
-
Antigenic and Polypeptide Structure of Turkey Enteric Coronaviruses as Defined by Monoclonal Antibodies
More LessSUMMARYTwenty-nine hybridoma cell lines, producing monoclonal antibodies (MAbs) to the Minnesota strain of turkey enteric coronavirus (TCV), have been established by fusion of Sp2/0 myeloma cells with spleen cells from BALB/c mice immunized with purified preparations of the egg-adapted or tissue culture-adapted virus. The hybridomas produced mainly IgG2a or IgG1 antibodies. Western immunoblotting experiments with purified virus, and immunoprecipitation tests with [35S]methionine-labelled infected cell extracts, allowed assessment of the polypeptide specificity of the MAbs. Sixteen hybridomas secreted antibodies directed to the peplomeric protein (E2, gp200/gp100) and putative intracellular precursors of apparent M r 170K to 180K and 90K. Four hybridomas produced antibodies that selectively reacted with a glycoprotein with an M r of 140K (E3). This polypeptide species corresponded to the major structural component of small granular projections, located near the base of the larger bulbous peplomers, and was found to be responsible for haemagglutination. The major neutralization-mediating determinants were found to be carried by both E2 and E3 glycoproteins. Eight hybridomas produced MAbs directed to the major nucleocapsid protein (N, 52K), and only one MAb reacted with a low M r structural glycoprotein (24K), corresponding to the matrix (El) protein. By indirect immunofluorescence, MAbs of different specificity also revealed distinct patterns of distribution of the viral antigens within the cells. The location on the virion of the antigenic determinants recognized by MAbs of different specificity was determined by the use of an immunogold electron microscopy technique. Comparison of nine TCV Quebec strains, using MAbs directed to peplomer and haemagglutinin proteins of the prototype Minnesota strain, confirmed their close antigenic relationship, but also revealed the occurrence of at least two distinct antigenic groups.
-
-
-
Several Rat Cell Lines Share a Common Defect in Their Inability to Internalize Murine Coronaviruses Efficiently
More LessSUMMARYInfection of rat cells, Schwannoma RN2, hepatoma HTC or myoblast L6, with the murine coronavirus JHM strain results in a persistent infection characterized by the release of virus over an extended period of time with a limited cytopathology. Several stages of the viral replication cycle have been examined in these cells in comparison to those in mouse L2 cells, which are totally permissive to JHM infection. Although the rat cells bound as much virus as the mouse cells their ability to internalize it was 40-fold less efficient than the mouse cells. This lower internalization efficiency was not enhanced by pH shock of infected cells, but was by treatment with polyethylene glycol. In all cell types there appeared to be no major differences in the ability of the internalized virus to replicate the viral RNA as determined by slot-blot analysis with a radiolabelled viral cDNA. A similar genetic mechanism appears to be operative in the lines because somatic cell hybrids formed between these lines in various combinations were also deficient in the ability to internalize bound virus. Taken together these results imply that rat cell lines in general share a common deficiency in their inability to internalize murine coronaviruses efficiently. This low efficiency in viral internalization may explain in part the ability of these lines to sustain persistent infections.
-
-
-
Monoclonal Antibodies to Bovine Coronavirus Glycoproteins E2 and E3: Demonstration of in vivo Virus-neutralizing Activity
More LessSUMMARYSix monoclonal antibodies (MAbs) to bovine coronavirus (BCV, Quebec isolate) E2 and E3 glycoproteins which were found previously to be neutralizing in vitro were examined for virus-neutralizing activity in vivo. Surgically ligated intestinal loops of newborn colostrum-deprived calves were virus-inoculated, mock-infected or inoculated with a mixture of virus and antibody. Of the six BCV-specific MAbs, four were found to be protective against a virulent field isolate of BCV, as indicated by a reduction in villous atrophy. These MAbs were specific to antigenic domain A and antigenic domains A1 and A2 on the E2 and E3 glycoproteins respectively. MAbs to antigenic domains B and C on the E2 and E3 glycoproteins, respectively, were not protective.
-
-
-
A Model for Persistent Murine Coronavirus Infection Involving Maintenance via Cytopathically Infected Cell Centres
More LessSUMMARYThe relatively cell impermeable hygromycin B was found to inhibit viral but not cellular protein synthesis when added to cultures of murine hepatitis virus (MHV)-infected or mock-infected mouse L-2 fibroblasts. Membrane permeability, as judged by influx of sodium ions, has previously been demonstrated to be an MHV E2 glycoprotein-mediated, cytopathic effect of MHV infection in L-2 cells. It is therefore likely that the selective effect of hygromycin B on viral protein synthesis is a reflection of an increased drug penetration into virus-infected cells. Using hygromycin B as a marker for MHV-induced cell membrane cytopathology, the effects of drug treatment on a persistent MHV infection in mouse LM-K fibroblasts were investigated. MHV persistence in LM-K cells, which normally involves a steady state infection of 0·1 to 1 % of the cells in culture, was found to be cured by hygromycin B treatment, as measured by the elimination of infectious virus from the supernatant medium. Hygromycin B also resulted in the eradication of MHV-specific RNA from LM-K cells, arguing against the presence of a non-cytopathically or latently infected subpopulation of cells.
-
-
-
Mapping of Neutralizing Epitopes to Fragments of the Bovine Coronavirus E2 Protein by Proteolysis of Antigen–Antibody Complexes
More LessSUMMARYNeutralizing antigenic domains on bovine coronavirus gp100/E2 were mapped to fragments of this protein by proteolytic cleavage and fragment analysis. The procedure involved analysis of fragments generated after incubation of E2–monoclonal antibody complexes with various proteases. The smallest antibody-bound fragments obtained were a 50K fragment following Staphylococcus aureus V8 protease and submaxillary protease digestion, and a 37K fragment following trypsin digestion. Trypsin also produced a transient antibody-bound 50K fragment. A 40K fragment which was not bound by antibody was observed following digestions with all three proteases. The 50K fragments generated by V8, submaxillary protease and trypsin comigrated on gels and displayed the same altered mobility under non-reducing conditions, suggesting identity of these fragments and indicating the presence of disulphide linkages in these fragments. The 40K fragments generated by these three enzymes also comigrated and displayed the same altered mobility under non-reducing conditions. The 37K trypsin fragment contained both neutralizing domains, A and B.
-
-
-
Synthesis and Processing of the Bovine Enteric Coronavirus Haemagglutinin Protein
More LessSUMMARYThe haemagglutinin molecule on the bovine enteric coronavirus has been identified as a glycoprotein of 140K composed of disulphide-linked subunits of 65K. In this study, we have shown the subunits to be identical by demonstrating an unambiguous amino-terminal amino acid sequence. The unglycosylated subunit was found to have an M r of 42·5K and to undergo rapid disulphide linkage and glycosylation. Glycosylation was found to be of the asparagine-linked type and some of the oligosaccharides underwent processing to complex forms. Studies with inhibitors of glycosylation suggested that a processing of the haemagglutinin oligosaccharide takes place on the virion whilst it is in the Golgi apparatus. Each haemagglutinin subunit on the mature virion was estimated to possess six or seven carbohydrate chains of either the high-mannose or hybrid type, and three or four chains of the complex type.
-
-
-
Cloning and in vitro Expression of the Gene for the E3 Haemagglutinin Glycoprotein of Bovine Coronavirus
More LessSUMMARYA cDNA clone representing the gene for the E3 glycoprotein, the haemagglutinin, of bovine coronavirus was isolated from a plasmid cDNA library of the viral genome and sequenced. The gene is located immediately 5′ of the E2 glycoprotein gene on the viral genome. Nucleotide sequencing of the E3 gene predicts a polypeptide of 424 amino acids with an M r of 47K. In vitro translation of mRNA transcribed from the cloned E3 gene yielded a polypeptide of M r 45K, similar to that predicted from the nucleotide sequence. In the presence of microsomal membranes, the in vitro product was cotranslationally processed to a 62K polypeptide which comigrated on SDS–polyacrylamide gels with the E3 monomer (gp62) obtained from virus-infected cells. Both the 45K and 62K polypeptides were immunoprecipitated with E3-specific monoclonal antibodies, confirming the identity of the gene as that encoding the E3 glycoprotein. Finally, only monoclonal antibodies to the E3 protein inhibited haemagglutination by the virus thus confirming its identity as the haemagglutinin of bovine coronavirus.
-
-
-
Antigenic Differentiation between Transmissible Gastroenteritis Virus of Swine and a Related Porcine Respiratory Coronavirus
More LessSummaryThe antigenic relationship between a recently isolated porcine respiratory coronavirus (TLM 83) and transmissible gastroenteritis (TGE) virus of swine was studied by neutralization, immunoblotting and radioimmunoassay (RIA) using TGE virus-specific monoclonal antibodies (MAbs) and polyclonal antibodies specific for both viruses. A complete two-way neutralization activity between the two viruses was found. Immunoblotting revealed cross-reactions between TLM 83 and TGE virus antigens at the level of the envelope protein (E1), the nucleoprotein (N) and the peplomer protein (E2). By virus neutralization assays and RIA with TGE virus-specific MAbs, the presence of similar epitopes in the E1 and N proteins and in the neutralization-mediating antigenic site of the E2 protein were demonstrated. E2 protein-specific MAbs, without neutralizing activity and reacting with antigenic sites B, C and D (previously defined), failed to recognize TLM 83. These results indicated a close antigenic relationship and structural similarity between TLM 83 and TGE viruses and also suggested potential ways of differentiating between the two viruses.
-
-
-
Identification of the Coronavirus MHV-JHM mRNA 4 Product
More LessSummaryA bacterial expression vector was constructed to encode a fusion protein which had, at its carboxy terminus, a polypeptide encoded within the 5′ proximal open reading frame of the coronavirus MHV-JHM mRNA 4. This polypeptide was isolated and used to produce an antiserum. The antiserum reacted specifically with a 15000 M r polypeptide synthesized in MHV-JHM-infected cells, or in vitro translations of infected cell poly(A) RNA enriched for mRNA 4. These results demonstrate the translational activity of mRNA 4 during infection, identify conclusively the translation product and provide a means to investigate the synthesis and function of this protein.
-
-
-
Evolution of Avian Coronavirus IBV: Sequence of the Matrix Glycoprotein Gene and Intergenic Region of Several Serotypes
More LessSummaryWe have sequenced 200 to 240 bases of the matrix (M) glycoprotein gene of 23 strains of infectious bronchitis virus (IBV) representing the A (D207), B (D3896), C (D3128), D (D212), Massachusetts (Mass), UK11 and UK12 serotypes. The bases examined code for the external, hydrophilic region and the first membrane-embedded hydrophobic region of M, both regions comprising approximately 20 amino acids. As predicted from protein M r studies the A/D and B/C serotypes had two and one potential glycosylation sites respectively. This variation appeared to derive from a combination of base substitutions and deletions/insertions. The glycosylation sequence Asn-Cys-Thr was highly conserved. Overall, the exposed part of M exhibited a fourfold greater extent of amino acid variation than did the membrane-embedded sequence. The transcription-associated homology region sequence (CUUAACAA) in the 5′ intergenic region was identical in all strains but there was considerable variation as to its location. The M gene of UK12 appeared to have evolved from a group A-like M gene by a two stage process involving a base substitution in the intergenic region which generated a new AUG translation start codon followed by deletion of the original AUG. Isolate UK11 closely resembled Mass strains in the intergenic region but was dissimilar from all strains in the protein coding region. The M sequences of serotypes B and C were identical and those of the A and D serotypes very similar. These results are discussed in relation to recent sequencing of part of the spike glycoprotein gene of some of these strains and the discovery of in vitro recombination of murine hepatitis coronavirus.
-
-
-
The Peplomer Protein E2 of Coronavirus JHM as a Determinant of Neurovirulence: Definition of Critical Epitopes by Variant Analysis
More LessSummaryWe selected murine coronavirus JHM variants specifically changed in defined antigenic sites of the peplomer protein E2. Variants were isolated from the supernatants of monoclonal antibody hybridoma cell cultures which continued to secrete neutralizing antibodies after being infected with JHM. Comparative antigenic analysis and biological tests were performed in order to refine an operational epitope map and to characterize functional domains important for pathogenicity. The reaction patterns (neutralization, inhibition of cell fusion, immunofluorescence and binding in ELISA) between the variant viruses and the panel of monoclonal antibodies were very similar. Four groups of variants were characterized each of which revealed distinct changes affecting one defined antigenic site. These observations indicated that at least four independently mutable antigenic sites were associated with domains involved in cell fusion, neutralization and pathogenicity (E2-Aa, -Ab, -Ba and -Bb). JHM variants with alterations in the E2-Aa, -Ab or -Bb sites were similar to wild-type virus. These variants caused acute hepatitis and encephalomyelitis in mice. In contrast, JHM variants with changes in site E2-Ba had a strong propensity to induce chronic disease accompanied by demyelination persisting for several months.
-
-
-
Structural Proteins of Bovine Coronavirus and Their Intracellular Processing
More LessSummaryThe Quebec isolate of bovine coronavirus (BCV) was found to contain four unique major structural proteins. These proteins consisted of the peplomeric protein (gp190/E2, gp100/E2), the nucleocapsid protein (p53/N) and its apparent trimer (p160/N), a family of small matrix glycoproteins (gp26/El, gp25/El and p23/El) and the putative haemagglutinin (gpl24/E3). Pulse-chase experiments utilizing polyclonal antiserum and monoclonal antibodies indicated that the unique BCV E3 protein had as its primary precursor an A-linked glycoprotein with an M r of 59000 (gp59) which underwent rapid dimerization by disulphide bond formation to yield gp118. Further glycosylation of gp118 produced gp124/E3 which incorporated fucose. Thus gp124/E3 was probably a homodimer. The processing of the E2 and E1 proteins of BCV was similar to that shown previously for mouse hepatitis virus. A large AM inked precursor glycoprotein, gpl70, underwent further glycosylation to yield gp190/E2 before subsequent proteolytic cleavage to yield gp100/E2. The glycosylated El (gp26, gp25) proteins arose as a result of O-linked glycosylation of p23/El as indicated by the resistance of these species to tunicamycin.
-
-
-
The Predicted Primary Structure of the Peplomer Protein E2 of the Porcine Coronavirus Transmissible Gastroenteritis Virus
More LessSummaryThe complete nucleotide sequence of cloned cDNAs containing the E2 glycoproteinencoding region of the genome of transmissible gastroenteritis virus (TGEV) has been determined. A single large translatable frame of 4·3 kb starting at 8·2 kb from the 3′ end of the genome was identified. Its deduced amino acid sequence contains the characteristic features of a coronavirus peplomer protein: (i) the precursor polypeptide of TGEV E2 is 1447 residues long (i.e. 285 longer than the avian infectious bronchitis coronavirus spike protein); (ii) partial N-terminal sequencing demonstrated that a putative secretory signal sequence of 16 amino acids is absent in the virion-associated protein; (iii) the predicted mol. wt. of the apoprotein is 158K; most of the 32 potential N-glycosylation sites available in the sequence are presumed to be functional to account for the difference between this and the experimentally determined value (200K to 220K); (iv) a typical hydrophobic sequence near the C terminus is likely to be responsible for anchoring the peplomer to the virion envelope.
-
-
-
Rat Glial C6 Cells are Defective in Murine Coronavirus Internalization
More LessSummaryRat C6 glial cells were resistant to infection by several strains of murine coronaviruses. The restriction was not at the adsorption stage, since virus adsorbed to the C6 cells in a similar manner to mouse L cells which supported a lytic infection. The virus could not be internalized by the C6 cells. However, if the virus was introduced into the C6 cells by polyethylene glycol fusion, viral replication occurred and progeny virions were released from the infected cells. These studies indicated that the C6 cells were restrictive to coronavirus replication by preventing the early penetration stage of the viral replicative cycle.
-
-
-
Sequence and N-terminal Processing of the Transmembrane Protein E1 of the Coronavirus Transmissible Gastroenteritis Virus
More LessSummarySequencing of part of a clone from a transmissible gastroenteritis virus genome cDNA library led to the identification of the gene encoding the E1 matrix protein. The amino acid sequence of the primary translation product predicts a polypeptide of 262 residues which shares many features with the previously characterized murine hepatitis virus and infectious bronchitis virus E1 proteins. However, N-terminal amino acid sequencing revealed that a putative signal peptide of 17 residues was absent in the virion-associated polypeptide. The predicted mol. wt. of the mature unglycosylated product, 27 800, is in agreement with the experimental M r value.
-
-
-
Intracellular RNAs of the Feline Infectious Peritonitis Coronavirus Strain 79-1146
More LessSUMMARYIn Felis catus whole foetus D cells infected with feline infectious peritonitis virus (FIPV), strain 79–1146, six virus-specific, poly(A)-containing RNA species of about 20, 9·6, 5·2, 3·8, 2·8 and 1·6 kb were found. By translation in vitro the 3·8 and 2·8 kb RNAs were shown to encode the 25K envelope protein and the 45K nucleocapsid protein, respectively. The partial map of the FIPV genome was compared with genomic maps of porcine, murine and avian coronaviruses. Differences in these maps suggest that transcription units have been lost or gained during coronavirus divergence.
-
-
-
Induction of Demyelination by a Temperature-sensitive Mutant of the Coronavirus MHV-A59 is Associated with Restriction of Viral Replication in the Brain
More LessSUMMARYThe neurovirulence of eight temperature-sensitive (ts) mutants of mouse hepatitis virus strain A59 in 4-week-old BALB/c mice was investigated. Whereas a dose of 100 p.f.u. of wild-type virus killed mice within a week, a 1000-fold higher dose of ts mutants did not. Three ts mutants induced demyelinating disease in the central nervous system (CNS). The pathology of the demyelinating disease caused by one mutant, designated ts-342, was studied in detail. Pathological changes, starting 3 days post-inoculation (p.i.), were characterized by inflammation and demyelination in the CNS. Antibody responses directed against all virus-specific structural proteins were present at 7 days p.i. No virus particles were observed by electron microscopy at 14 days p.i. However, macrophages and lymphocytes were abundant in the areas of demyelination. The growth kinetics in vivo of wild-type virus, ts-342 and a revertant of ts-342 were compared. Wild-type virus and the revertant replicated rapidly in the brain and spread to the liver causing a lethal hepatitis. Ts-342, however, replicated to a much lesser extent within the brain and could not be detected in the blood or liver. The ts lesion in the genome of ts-342 seems, therefore, to determine the outcome of the infection.
-
-
-
Completion of the Sequence of the Genome of the Coronavirus Avian Infectious Bronchitis Virus
More LessSUMMARYThe nucleotide sequence determination of the genome of the Beaudette strain of the coronavirus avian infectious bronchitis virus (IBV) has been completed. The complete sequence has been obtained from 17 overlapping cDNA clones, the 5′-most of which contains the leader sequence (as determined by direct sequencing of the genome) and the 3′-most of which contains the poly(A) tail. Approximately 8 kilobases at the 3′ end of this sequence have already been published. These contain the sequences of mRNAs A to E within which are the genes for the spike, the membrane and the nucleocapsid polypeptides: the main structural components of the virion. The remainder of the sequence, equivalent to the ‘unique’ region of mRNA F, is some 20 kilobases in length and is thought to code for a polymerase or polymerases which are involved in the replication of the genome and the production of the subgenomic messenger RNAs. This sequence contains two large open reading frames, potentially coding for polypeptides of molecular weights 441000 and 300000. Unlike other large open reading frames in the virus, the 300000 open reading frame appears to have no subgenomic RNA associated with it which would allow it to be at the 5′ end of an mRNA species. Because of this, and because of the characteristics of the sequence in the region immediately upstream of its start codon, other mechanisms of translation, such as ribosome slippage, must be postulated.
-
-
-
Nucleotide Sequence of the Gene Encoding the Surface Projection Glycoprotein of Coronavirus MHV-JHM
More LessSUMMARY
Sequences encoding the surface projection glycoprotein of the coronavirus, murine hepatitis virus (MHV), strain JHM, have been cloned into pAT153 using cDNA produced by priming with specific oligonucleotides on infected cell RNA. The regions of three clones pJMS1010, pJS112 and pJS92, which together encompass the surface protein gene have been sequenced by the chain termination method. The sequence of the primary translation product, deduced from the DNA sequence, predicts a polypeptide of 1235 amino acids with a molecular weight of 136600. This polypeptide displays the features characteristic of a group 1 membrane protein; an amino-terminal signal sequence and carboxy-terminal membrane and cytoplasmic domains. There are 21 potential glycosylation sites in the polypeptide and a cysteine-rich region in the vicinity of the transmembrane domain. During maturation proteolytic processing of the polypeptide occurs and at positions 624 to 628 the sequence Arg-Arg-Ala-Arg- Arg is found, which is similar to a number of basic sequences involved in the cleavage of enveloped RNA virus glycoproteins. The fusogenic properties of the MHV surface protein do not appear to correlate with a strongly hydrophobic region at the putative amino terminus of the carboxy-terminal cleavage product.
-
-
-
Comparison of the Spike Precursor Sequences of Coronavirus IBV Strains M41 and 6/82 with that of IBV Beaudette
More LessSummaryThe nucleotide sequences of the spike precursor genes of infectious bronchitis virus strains M41 and 6/82 have been determined and compared with that of the Beaudette strain which we have previously sequenced. The two Massachusetts strains, M41 and Beaudette, were found to be remarkably similar, having only 3.7% of the amino acids different. The situation with 6/82, one of the new field isolates, is quite different and this strain had 13.8% of its amino acids different from Beaudette. The differences identified are discussed in terms of the structural features of the spike protein.
-
-
-
Antigenic Structure of Transmissible Gastroenteritis Virus. II. Domains in the Peplomer Glycoprotein
More LessSummaryThe antigenic structure of the peplomer glycoprotein E2 of the porcine transmissible gastroenteritis coronavirus (TGEV) was explored using a panel of 23 hybridoma antibodies (MAbs). The topography of the epitopes was established by means of a competition radioimmunoassay. Four main antigenic sites, termed A, B, C and D, were thus clearly delineated. Most of the neutralization-mediating determinants were found to cluster in the A-B area, which has been shown to be highly conserved among TGEV strains. Cooperative enhancement of binding to sites B and D was observed following attachment of MAbs relevant to site A. Additional epitopes were identified on E2 by MAbs that selectively recognized its intracellular precursor. Functional mapping was also performed using neutralization-resistant variants. Analysis of their reactivity confirmed part of the epitope linkages defined by the first approach. The overall lower frequency of such variants altered at site A suggested that some of the epitopes may play an essential function.
-
-
-
Coronavirus IBV: Removal of Spike Glycopolypeptide S1 by Urea Abolishes Infectivity and Haemagglutination but Not Attachment to Cells
More LessSummaryUrea has been used to remove the S1 spike glycopolypeptide from avian infectious bronchitis virus (IBV) strains M41 and Beaudette, without removing the S2 spike-anchoring glycopolypeptide. Reduction of the pH to 2.9 did not cause release of S1 although some S1 was released spontaneously from IBV Beaudette at pH 7.4. Virus that lacked S1 was no longer infectious or able to cause haemagglutination (HA). However, radiolabelled IBV that lacked S1 attached to erythrocytes and chick kidney cells to the same or similar extent as did intact virus. Treatment of IBV with a phospholipase C preparation, required to make IBV cause HA, did not increase binding of IBV to erythrocytes. The results indicate that while the attachment to cells of virus that lacks S1 is qualitatively different from that of intact virus, the decline in infectivity is the consequence of the loss of some other spike function.
-
