 -
Volume 147,
Issue 2,
2001
-
Volume 147,
Issue 2,
2001
Volume 147, Issue 2, 2001
- Review Article
-
- Microbiology Comment
-
- Biochemistry
-
-
-
The glyceraldehyde-3-phosphate dehydrogenase polypeptides encoded by the Saccharomyces cerevisiae TDH1, TDH2 and TDH3 genes are also cell wall proteins
More LessThe authors show that the glycolytic enzyme glyceraldehyde-3-phosphate dehydrogenase (GAPDH) of Saccharomyces cerevisiae, previously thought to be restricted to the cell interior, is also present in the cell wall. GAPDH activity, proportional to cell number and time of incubation, was detected in intact wild-type yeast cells. Intact cells of yeast strains containing insertion mutations in each of the three structural TDH genes (tdh1, tdh2 and tdh3) and double mutants (tdh1 tdh2 and tdh1 tdh3) also displayed a cell-wall-associated GAPDH activity, in the range of parental wild-type cells, although with significant differences among strains. A cell wall location of GAPDH was further confirmed in wild-type and tdh mutants by indirect immunofluorescence and flow cytometry analysis with a polyclonal antibody against S. cerevisiae GAPDH. By immunoelectron microscopy, the GAPDH protein was detected at the outer surface of the cell wall of wild-type cells, as well as in the cytoplasm. Western immunoblot analysis of cell wall extracts and cytosol showed that Tdh2 and Tdh3 polypeptides are present in the cell wall, as well as in the cytosol, of exponentially growing cells. Tdh1 is only detected in stationary-phase cells, again in both cytosol and cell wall extracts. The results incorporate the GAPDH of S. cerevisiae, encoded by TDH1–3, into the newly emerging family of multifunctional cell-wall-associated GAPDHs which retain their catalytic activity.
-
-
-
-
Frankia sequences exhibiting RNA polymerase promoter activity
More LessThe GenBank accession number for the sequence reported in this paper is AY008259.
Frankia are Gram-positive, filamentous bacteria capable of fixing atmospheric dinitrogen either in the free-living state or in symbiosis with a variety of woody plants. Only a few Frankia genes have been sequenced and gene expression is not well characterized. To isolate a segment of Frankia DNA that functions as an RNA polymerase promoter, fragments of Frankia strain ArI5 genomic DNA were cloned upstream of a promoterless, Vibrio harveyi luxAB cassette. Constructs were screened for luminescence in E. coli and positive clones assayed for in vitro transcription activity with a partially purified Frankia RNA polymerase extract. Primer extension analysis of in vitro transcripts produced from one clone, GLO7, identified two major transcription start sites, TSP-1 and TSP-2, 52 bp apart. Deletion analysis then localized sequences essential for promoter activity. The upstream promoter region, GLO7p1, contains sequences resembling the −35 element of a Streptomyces promoter and the −35 and −10 elements of the canonical E. coli promoter. Also within this region are two pentamers identical to sequences near the 5′ end of the Frankia strain CpI1 glutamine synthetase gene. The second promoter, GLO7p2, contains a putative NtrC binding site at −145 and a possible σN-RNA polymerase recognition sequence at −14 suggesting that GLO7p2 may be a nitrogen-regulated promoter. An in vivo transcript representing an ORF of 498 aa starting 64 bp downstream of the distal transcription start, TSP-1, was detected by RT-PCR. This supports the conclusion that this DNA fragment has promoter activity in vivo as well as in vitro.
-
- Development And Structure
-
-
-
Assessment of lectin-binding analysis for in situ detection of glycoconjugates in biofilm systems
More LessAn assessment of lectin-binding analysis for the characterization of extracellular glycoconjugates as part of the extracellular polymeric substances in environmental microbial communities was performed using fully hydrated river biofilms. The applicability of the method was evaluated for single, dual and triple staining with a panel of fluor-conjugated lectins. It was shown that lectin-binding analysis was able to stain glycoconjugates within biofilm communities. Lectin staining also demonstrated spatial heterogeneity within the biofilm matrix. Furthermore, the application of two or even three lectins was possible if suitable combinations were selected. The lectin-binding analysis can be combined with general nucleic acid stains to collect both nucleic acid and glycoconjugate signals. The effects of incubation time, lectin concentration, fluor labelling, carbohydrate inhibition, order of addition and lectin interactions were studied. An incubation time of 20 min was found to be sufficient for completion of lectin binding. It was not possible to ascertain saturating concentration for individual lectins, therefore a standard concentration was used for the assay. Carbohydrate inhibition tests indicated that fluorescein isothiocyanate (FITC)-conjugated lectins had more specific binding characteristics than tetramethyl rhodamine isothiocyanate (TRITC)- or cyanine dye (CY5)-labelled lectins. The order of addition and the nature of the fluor conjugate were also found to influence the binding pattern of the lectins. Therefore the selection of a panel of lectins for investigating the EPS matrix must be based on a full evaluation of their behaviour in the biofilm system to be studied. Despite this necessity, lectin-binding analysis represents a valuable tool to examine the glycoconjugate distribution in fully hydrated biofilms. Thereby, chemical heterogeneities within extracellular biofilm locations can be identified in order to examine the role (e.g. sorption properties, microenvironments, cell–extracellular polymeric subtance interactions) of the extracellular polymeric substances in environmental biofilm systems.
-
-
- Environmental Microbiology
-
-
-
Characterization of microbial consortia in a terephthalate-degrading anaerobic granular sludge system
More LessThe GenBank accession numbers for the sequences obtained in this work are AF229774–AF229793.
The microbial composition and spatial distribution in a terephthalate-degrading anaerobic granular sludge system were characterized using molecular techniques. 16S rDNA clone library and sequence analysis revealed that 78·5% of 106 bacterial clones belonged to the δ subclass of the class Proteobacteria; the remaining clones were assigned to the green non-sulfur bacteria (7·5%), Synergistes (0·9%) and unidentified divisions (13·1%). Most of the bacterial clones in the δ-Proteobacteria formed a novel group containing no known bacterial isolates. For the domain Archaea, 81·7% and 18·3% of 72 archaeal clones were affiliated with Methanosaeta and Methanospirillum, respectively. Spatial localization of microbial populations inside granules was determined by transmission electron microscopy and fluorescent in situ hybridization with oligonucleotide probes targeting the novel δ-proteobacterial group, the acetoclastic Methanosaeta, and the hydrogenotrophic Methanospirillum and members of Methanobacteriaceae. The novel group included at least two different populations with identical rod-shape morphology, which made up more than 87% of the total bacterial cells, and were closely associated with methanogenic populations to form a nonlayered granular structure. This novel group was presumed to be the primary bacterial population involved in the terephthalate degradation in the methanogenic granular consortium.
-
-
- Genetics And Molecular Biology
-
-
-
Identification of cell wall proteins of Bacteroides fragilis to which bacteriophage B40-8 binds specifically
More LessThe PIR accession numbers for the sequences reported in this paper are A59325 (BactA) and B59325 (BactB).
Bacteriophage infecting Bacteroides fragilis, one of the most abundant bacteria in the human colon, have been proposed as indicators of virological faecal pollution. The first identification of a receptor for a bacteriophage in B. fragilis is reported here. First, resistant mutants were characterized following phage inactivation, and it was shown that cell wall proteins are involved in phage binding. Then the proteins involved were identified by various approaches: (i) comparison of the protein profiles of wild-type B. fragilis HSP40 and phage-resistant mutants; (ii) application of a modification of the virus overlay protein blot assay (VOPBA). At least two proteins of B. fragilis, with apparent molecular masses of 35 ± 5 kDa and 65 ± 5 kDa, bind to B40-8. This result was later confirmed by running a complex consisting of this phage bound to radiolabelled proteins of B. fragilis on an immunoaffinity column loaded with a specific antibody against the phage. Cell proteins retained in the column also coincided with the proteins that differed in the profiles of resistant mutants. Finally, to identify the potential function of these two proteins, their N-terminal sequences were determined and compared to published sequences, but no homologies were found.
-
-
-
-
Properties of the major non-specific endonuclease from the strict anaerobe Fibrobacter succinogenes and evidence for disulfide bond formation in vivo
More LessDNase A is a non-specific endonuclease of Fibrobacter succinogenes. The enzyme was purified to homogeneity and its properties studied both in vitro and in vivo. Magnesium but not calcium was essential for nucleolytic activity. Manganese ions substituted for magnesium but were less stimulatory. DNase A activity was markedly inhibited by either NaCl or KCl at concentrations greater than 75 mM. The enzyme had a temperature optimum of 25 °C and a pH optimum of about 7·0. Values for K m and K cat were determined to be 61 μM and 330 s−1 respectively, with a catalytic efficiency approximately threefold greater than bovine pancreatic DNase I, but 10-fold less than the Serratia marcescens NucA. DNase A was localized to the periplasm and probably exists as a monomeric species. The enzyme possessed one or more disulfide bonds. In the reduced form it had an apparent mass of 33 kDa, while in the oxidized form it was 29 kDa as estimated by SDS-PAGE. Reduction of the disulfide bonds by dithiothreitol with or without subsequent alkylation by iodoacetamide strongly inactivated the enzyme. DNase A accumulated in vivo had an apparent mass of 29 kDa, indicating that it was in an oxidized form. This is the first indication in a strict anaerobe of a functional periplasmic disulfide bond forming system, phenotypically similar to Dsb systems in facultative and aerobic bacteria.
-
-
-
Construction and analysis of β-lactamase-inhibitory protein (BLIP) non-producer mutants of Streptomyces clavuligerus
More LessThe GenBank accession number for the sequence determined in this work is M34538.
The gene encoding BLIP, a beta-lactamase-inhibitory protein, was disrupted in wild-type Streptomyces clavuligerus and in a clavulanic acid non-producing mutant. The resulting BLIP mutant and BLIP/clavulanic acid double mutant showed no residual proteinaceous β-lactamase-inhibitory activity, indicating that only a single β-lactamase-inhibitory protein exists in S. clavuligerus. The lack of any proteinaceous β-lactamase-inhibitory activity in the bli and bli/claR mutants also indicates that BLP, the BLIP-like protein, encoded by S. clavuligerus does not possess β-lactamase-inhibitory activity despite its similarity to BLIP. The bli mutant and the bli/claR double mutant did not show any aberrant growth morphology, sporulation defects, or alterations in cephamycin C production or penicillin G resistance when compared to wild-type S. clavuligerus or to the claR single mutant. Mutants bearing the bli gene disruption did show an elevated level of production of clavam-2-carboxylate and hydroxymethyl clavam as well as clavulanic acid. This phenomenon was observed in the middle stages of production of these clavams but was not detected during maximum production. The production of BLIP was also determined to be down-regulated in a ccaR mutant, lacking the pathway-specific transcriptional regulator required for production of cephamycin C and clavulanic acid. Sequencing of the regions flanking the bli gene showed the presence of a partial open reading frame that encodes a DNA-binding protein, and several open reading frames apparently involved in the production of an ABC transporter.
-
-
-
A novel rolling-circle-replicating plasmid from Pseudomonas putida P8: molecular characterization and use as vector
More LessThe EMBL and GenBank accession number for the nucleotide sequence of pPP8–1 is AJ289784.
In Pseudomonas putida P8, three cryptic circular plasmids were detected, i.e. pPP8-1 (2·5 kbp), pPP8-2 (42 kbp) and pPP8-3 (
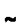 100 kbp). Cloning and complete sequencing of pPP8-1 revealed a 2534 bp element harbouring four open reading frames (ORFs A, B, C and D). No function could be attributed to the latter three ORFs, whereas the predicted ORF A gene product is homologous to replication proteins known from small multicopy plasmids of Gram-positive bacteria and single-stranded (ss) phages, genetic elements replicating via a rolling circle (RC) mechanism involving characteristic ssDNA intermediates. Consistently, a double-strand origin of replication, highly conserved in rolling-circle-replicating (RCR) elements, was identified in pPP8-1, along with a putative single-strand origin. Beyond this, ss replication intermediates were confirmed by Southern analysis and mungbean-nuclease digestion. This being the first element of this type known in pseudomonads, a kanamycin-resistance gene was ligated into pPP8-1 and the resulting vector was successfully used for the transformation of both Escherichia coli and P. putida.
100 kbp). Cloning and complete sequencing of pPP8-1 revealed a 2534 bp element harbouring four open reading frames (ORFs A, B, C and D). No function could be attributed to the latter three ORFs, whereas the predicted ORF A gene product is homologous to replication proteins known from small multicopy plasmids of Gram-positive bacteria and single-stranded (ss) phages, genetic elements replicating via a rolling circle (RC) mechanism involving characteristic ssDNA intermediates. Consistently, a double-strand origin of replication, highly conserved in rolling-circle-replicating (RCR) elements, was identified in pPP8-1, along with a putative single-strand origin. Beyond this, ss replication intermediates were confirmed by Southern analysis and mungbean-nuclease digestion. This being the first element of this type known in pseudomonads, a kanamycin-resistance gene was ligated into pPP8-1 and the resulting vector was successfully used for the transformation of both Escherichia coli and P. putida.
-
-
-
The aprX–lipA operon of Pseudomonas fluorescens B52: a molecular analysis of metalloprotease and lipase production
More LessThe GenBank accession numbers for the sequences reported in this paper are AF216700, AF216701 and AF216702.
Extracellular protease and lipase production by psychrotrophic strains of Pseudomonas fluorescens is repressed by iron and regulated by temperature. The regulation of protease and lipase has been investigated in P. fluorescens B52. Whereas lipase production is increased below the optimum growth temperature (‘low-temperature regulation’), protease production was relatively constant and only decreased above the optimum growth temperature. The genes encoding protease (aprX) and lipase (lipA) are encoded at opposite ends of a contiguous set of genes which also includes protease inhibitor, Type I secretion functions and two autotransporter proteins. Evidence is presented indicating that these genes constitute an operon, with a promoter adjacent to aprX which has been identified by S1 nuclease analysis. The regulation of aprX and lipA has been investigated at the RNA level and using lacZ fusion strains. Whereas the data are consistent with iron regulation at the transcriptional level, a lipA′–′lacZ fusion is not regulated by temperature, suggesting that temperature regulation is post-transcriptional or post-translational. The possibility of regulation at the level of mRNA decay is discussed.
-
-
-
The Pseudomonas fluorescens transcription activator AdnA is required for adhesion and motility
More LessThe GenBank accession number for the sequence reported in this paper is AF312695.
The locations of two mutations that prevent adhesion of Pseudomonas fluorescens Pf0-1 to sand columns and seeds (adn, adhesion) were identified. Both lie in a single gene showing homology to the NtrC/NifA family of transcription activators. The predicted 55 kDa protein encoded by adnA is most closely related to activators involved in expression of flagellar proteins, consistent with the lack of flagella in adnA strains. Constitutive adnA expression restored motility and adhesion to an adnA strain, demonstrating that the observed phenotypes are due to lack of AdnA and not a consequence of other mutations or polar effects of mutations in adnA on other genes.
-
-
-
A soluble two-component regulatory system controls expression of quinoprotein ethanol dehydrogenase (QEDH) but not expression of cytochrome c 550 of the ethanol-oxidation system in Pseudomonas aeruginosa
More LessThe GenBank accession number for the sequence reported in this work is AJ009858.2 (=CAB 95009.1).
The regulation of the divergent promoters of the exaAB genes in Pseudomonas aeruginosa ATCC 17933, in which exaA encodes a quinoprotein ethanol dehydrogenase and exaB codes for a cytochrome c 550, was studied. Using transcriptional lacZ fusions, promoter activity during growth on several substrates was measured. These promoter-probe vectors were also used to identify regulatory mutants defective in exaAB induction. Transcription from both exaA and exaB was reduced significantly in four mutants. Two other mutants showed transcription from exaA that was reduced, but higher than wild-type transcription from exaB. The genes that are needed for exaA promoter induction were sequenced and found to encode a two-component regulatory system: a histidine sensor kinase, which lacks a transmembrane helical N-terminus and is presumably located in the cytoplasm, and a response regulator. The phenotypic characterization and restoration of the wild-type behaviour of the different regulatory mutants produced by different cosmids and subclones indicate that six different genes may be involved in regulating ethanol oxidation in P. aeruginosa.
-
-
-
Species-specific inhibition of fungal protein synthesis by sordarin: identification of a sordarin-specificity region in eukaryotic elongation factor 2
The GenBank accession numbers for the sequences reported in this manuscript are AF107286–AF107291, AF292693 and AF248644.
More LessThe sordarin class of natural products selectively inhibits fungal protein synthesis by impairing the function of eukaryotic elongation factor 2 (eEF2). Mutations in Saccharomyces cerevisiae eEF2 or the ribosomal stalk protein rpP0 can confer resistance to sordarin, although eEF2 is the major determinant of sordarin specificity. It has been shown previously that sordarin specifically binds S. cerevisiae eEF2 while there is no detectable binding to eEF2 from plants or mammals, despite the high level of amino acid sequence conservation among these proteins. In both whole-cell assays and in vitro translation assays, the efficacy of sordarin varies among different species of pathogenic fungi. To investigate the basis of sordarin’s fungal selectivity, eEF2 has been cloned and characterized from several sordarin-sensitive and -insensitive fungal species. Results from in vivo expression of Candida species eEF2s in S. cerevisiae and in vitro translation and growth inhibition assays using hybrid S. cerevisiae eEF2 proteins demonstrate that three amino acid residues within eEF2 account for the selectivity of this class of compounds. It is also shown that the corresponding residues at these positions in human eEF2 are sufficient to confer sordarin insensitivity to S. cerevisiae identical to that observed with mammalian eEF2.
-
-
-
Streptococcus pyogenes sclB encodes a putative hypervariable surface protein with a collagen-like repetitive structure
More LessThe GenBank accession numbers for the sequences determined in this work are given in the text and Table 1 T1 .
Streptococcus pyogenes is the causative agent in a wide range of diseases of humans of varying severity. During a study scanning the genome sequence of a serotype M1 invasive isolate SF370 for novel surface proteins, an ORF, designated sclB, was identified. The putative protein encoded by sclB contains both a signal peptide and classic Gram-positive wall-associated sequences. Comparison of the sequences of this ORF with those from a number of unrelated isolates demonstrated that sclB encodes a putative surface protein with a variable N-terminal sequence followed by a variable length tract of collagen-like GXYn repeats. A further feature of sclB is the presence of CAAAA repeat tracts immediately downstream of the putative start codon. The number of these pentameric repeats varies from 4 to 15 between strains and variation in repeat number results in the predicted SclB protein being either in or out of frame relative to the start codon. These observations suggest that expression of this protein may be regulated at the translational level as a result of gain or loss of CAAAA repeats. While the function of SclB remains to be elucidated, an sclB-specific transcript was detected by RT-PCR during in vitro culture. Finally, it is shown that a second gene, sclA, potentially encoding a protein with a similar extensive collagen-like structure and variable N-terminal sequence, is present in all isolates of S. pyogenes tested to date. Thus S. pyogenes harbours a novel family of structurally related and surface-exposed proteins of potential importance in the pathogenic process.
-
- Pathogenicity And Medical Microbiology
-
-
-
Environmentally regulated genes of Streptococcus suis: identification by the use of iron-restricted conditions in vitro and by experimental infection of piglets
The GenBank accession numbers for the sequences determined in this work are AF302190–AF302207 (iri genes) and AF303226–303247 (ivs genes).
More LessThe identification of environmentally regulated genes of Streptococcus suis by the use of iron-restricted conditions in vitro and by experimental infection of piglets is described. Eighteen unique iron-restriction-induced (iri) genes and 22 unique in-vivo-selected (ivs) genes of Strep. suis were found. None of the ivs genes was exclusively expressed in vivo. Four iri genes were identical to four clones selected in piglets. Two ivs genes were similar to genes for putative virulence factors. One of these ivs genes was identical to the epf gene of virulent Strep. suis serotype 2 strains and the other showed homology to a gene encoding a fibronectin-binding protein of Streptococcus gordonii. Two additional ivs genes showed homology to environmentally regulated genes previously identified by using an in vivo expression technology (IVET) selection system in other bacterial species. One of these showed similarity to the agrA gene of Staphylococcus aureus, a key locus involved in the regulation of numerous virulence proteins. The promoter selection system described in this paper has been successfully used for the identification of many environmentally regulated genes potentially involved in the pathogenesis of Strep. suis infections in piglets.
-
-
-
-
Characterization of the Streptococcus pneumoniae NADH oxidase that is required for infection
More LessStreptococcus pneumoniae is an important human pathogen capable of causing serious infections. NADH oxidase, a factor necessary for infection, was previously identified as part of a signature-tagged mutagenesis screen of a S. pneumoniae clinical isolate, 0100993. The mutant, with a plasmid insertion disrupting the nox gene, was attenuated for virulence in a murine respiratory tract infection model. A complete refined nox deletion mutant was generated by allelic-replacement mutagenesis and found to be attenuated for virulence 105-fold in the murine respiratory tract infection model and at least 104-fold in a Mongolian gerbil otitis media infection model, confirming the importance of the NADH oxidase for both types of S. pneumoniae infection. NADH oxidase converts O2 to H2O. If O2 is not fully reduced, it can form superoxide anion () and hydrogen peroxide (H2O2), both of which can be toxic to cells. Bacterial cell extracts from the allelic-replacement mutant were found to lack NADH oxidase activity and the mutant was unable to grow exponentially under conditions of vigorous aeration. In contrast, the mutant displayed normal growth characteristics under conditions of limited aeration. The S. pneumoniae nox gene was cloned and expressed in E. coli. The purified His-tagged NADH oxidase was shown to oxidize NADH with a K m of 32 μM, but was unable to oxidize NADPH. Oxidation of NADH was independent of exogenous FAD or FMN.
-
Volumes and issues
-
Volume 170 (2024)
-
Volume 169 (2023)
-
Volume 168 (2022)
-
Volume 167 (2021)
-
Volume 166 (2020)
-
Volume 165 (2019)
-
Volume 164 (2018)
-
Volume 163 (2017)
-
Volume 162 (2016)
-
Volume 161 (2015)
-
Volume 160 (2014)
-
Volume 159 (2013)
-
Volume 158 (2012)
-
Volume 157 (2011)
-
Volume 156 (2010)
-
Volume 155 (2009)
-
Volume 154 (2008)
-
Volume 153 (2007)
-
Volume 152 (2006)
-
Volume 151 (2005)
-
Volume 150 (2004)
-
Volume 149 (2003)
-
Volume 148 (2002)
-
Volume 147 (2001)
-
Volume 146 (2000)
-
Volume 145 (1999)
-
Volume 144 (1998)
-
Volume 143 (1997)
-
Volume 142 (1996)
-
Volume 141 (1995)
-
Volume 140 (1994)
-
Volume 139 (1993)
-
Volume 138 (1992)
-
Volume 137 (1991)
-
Volume 136 (1990)
-
Volume 135 (1989)
-
Volume 134 (1988)
-
Volume 133 (1987)
-
Volume 132 (1986)
-
Volume 131 (1985)
-
Volume 130 (1984)
-
Volume 129 (1983)
-
Volume 128 (1982)
-
Volume 127 (1981)
-
Volume 126 (1981)
-
Volume 125 (1981)
-
Volume 124 (1981)
-
Volume 123 (1981)
-
Volume 122 (1981)
-
Volume 121 (1980)
-
Volume 120 (1980)
-
Volume 119 (1980)
-
Volume 118 (1980)
-
Volume 117 (1980)
-
Volume 116 (1980)
-
Volume 115 (1979)
-
Volume 114 (1979)
-
Volume 113 (1979)
-
Volume 112 (1979)
-
Volume 111 (1979)
-
Volume 110 (1979)
-
Volume 109 (1978)
-
Volume 108 (1978)
-
Volume 107 (1978)
-
Volume 106 (1978)
-
Volume 105 (1978)
-
Volume 104 (1978)
-
Volume 103 (1977)
-
Volume 102 (1977)
-
Volume 101 (1977)
-
Volume 100 (1977)
-
Volume 99 (1977)
-
Volume 98 (1977)
-
Volume 97 (1976)
-
Volume 96 (1976)
-
Volume 95 (1976)
-
Volume 94 (1976)
-
Volume 93 (1976)
-
Volume 92 (1976)
-
Volume 91 (1975)
-
Volume 90 (1975)
-
Volume 89 (1975)
-
Volume 88 (1975)
-
Volume 87 (1975)
-
Volume 86 (1975)
-
Volume 85 (1974)
-
Volume 84 (1974)
-
Volume 83 (1974)
-
Volume 82 (1974)
-
Volume 81 (1974)
-
Volume 80 (1974)
-
Volume 79 (1973)
-
Volume 78 (1973)
-
Volume 77 (1973)
-
Volume 76 (1973)
-
Volume 75 (1973)
-
Volume 74 (1973)
-
Volume 73 (1972)
-
Volume 72 (1972)
-
Volume 71 (1972)
-
Volume 70 (1972)
-
Volume 69 (1971)
-
Volume 68 (1971)
-
Volume 67 (1971)
-
Volume 66 (1971)
-
Volume 65 (1971)
-
Volume 64 (1970)
-
Volume 63 (1970)
-
Volume 62 (1970)
-
Volume 61 (1970)
-
Volume 60 (1970)
-
Volume 59 (1969)
-
Volume 58 (1969)
-
Volume 57 (1969)
-
Volume 56 (1969)
-
Volume 55 (1969)
-
Volume 54 (1968)
-
Volume 53 (1968)
-
Volume 52 (1968)
-
Volume 51 (1968)
-
Volume 50 (1968)
-
Volume 49 (1967)
-
Volume 48 (1967)
-
Volume 47 (1967)
-
Volume 46 (1967)
-
Volume 45 (1966)
-
Volume 44 (1966)
-
Volume 43 (1966)
-
Volume 42 (1966)
-
Volume 41 (1965)
-
Volume 40 (1965)
-
Volume 39 (1965)
-
Volume 38 (1965)
-
Volume 37 (1964)
-
Volume 36 (1964)
-
Volume 35 (1964)
-
Volume 34 (1964)
-
Volume 33 (1963)
-
Volume 32 (1963)
-
Volume 31 (1963)
-
Volume 30 (1963)
-
Volume 29 (1962)
-
Volume 28 (1962)
-
Volume 27 (1962)
-
Volume 26 (1961)
-
Volume 25 (1961)
-
Volume 24 (1961)
-
Volume 23 (1960)
-
Volume 22 (1960)
-
Volume 21 (1959)
-
Volume 20 (1959)
-
Volume 19 (1958)
-
Volume 18 (1958)
-
Volume 17 (1957)
-
Volume 16 (1957)
-
Volume 15 (1956)
-
Volume 14 (1956)
-
Volume 13 (1955)
-
Volume 12 (1955)
-
Volume 11 (1954)
-
Volume 10 (1954)
-
Volume 9 (1953)
-
Volume 8 (1953)
-
Volume 7 (1952)
-
Volume 6 (1952)
-
Volume 5 (1951)
-
Volume 4 (1950)
-
Volume 3 (1949)
-
Volume 2 (1948)
-
Volume 1 (1947)
Most Read This Month


