 -
Volume 147,
Issue 2,
2001
-
Volume 147,
Issue 2,
2001
Volume 147, Issue 2, 2001
- Pathogenicity And Medical Microbiology
-
-
-
Comparison of toxinotyping and PCR ribotyping of Clostridium difficile strains and description of novel toxinotypes
More LessToxinotyping and PCR ribotyping are two methods that have been used to type Clostridium difficile isolates. Toxinotyping is based on PCR-RFLP analysis of a 19 kb region encompassing the C. difficile pathogenicity locus. PCR ribotyping is based on comparison of patterns of PCR products of the 16S–23S rRNA intergenic spacer region. Representative strains (101) from a C. difficile PCR ribotype library and 22 strains from previously described toxinotypes were analysed to compare ribotyping with toxinotyping. Within this panel of strains all 11 toxinotypes (0–X) described previously and an additional 5 novel toxinotypes (XI–XV) were observed. PCR ribotyping and toxinotyping correlated well and usually all strains within a given ribotype had similar changes in toxin genes. The new toxinotype XI comprises strains that did not express toxins TcdA or TcdB at detectable levels, but contained part of the tcdA gene. Strains of toxinotype XII exhibit changes only in the 5′ end of the tcdB gene. Toxinotype XIV is composed of strains that have a large insertion at the beginning of the tcdA gene. A total of 25 of the 89 tested PCR ribotypes of C. difficile contained variant strains. It was estimated that they represent 7·7% of the total number of strains in the Anaerobe Reference Unit collection.
-
-
-
-
Edwardsiella tarda mutants defective in siderophore production, motility, serum resistance and catalase activity
More LessThe GenBank accession numbers for the sequences determined in this work are AF324338–AF324342 (Table 3 T3 ).
Edwardsiella tarda is a Gram-negative bacterium that causes a systemic infection, edwardsiellosis, in fish. The virulence factors of this pathogen and its genetic determinants have not been systematically examined. In this study, TnphoA transposon mutagenesis was used to construct a library of 440 alkaline phosphatase (PhoA+) fusion mutants from a total of 400000 transconjugants derived from Ed. tarda PPD130/91. This library included genes for secreted and membrane-associated proteins normally involved in virulence. The library was screened for four virulence factors: siderophore production, motility, serum resistance and catalase production. Eight mutants deficient in one or more of these phenotypes were grouped into four classes. They were further characterized for their stimulation of reactive oxygen intermediate production by fish phagocytes, for their adhesion to and internalization into EPC (epithelioma papillosum of carp) cells, and for attenuation of virulence in blue gourami. Mutants 2A and 34 were highly attenuated in fish, with LD50 values about 10 times higher than for the wild-type. These strains had mutations in the genes encoding arylsulfate sulfotransferase (mutant 2A) and a catalase precursor protein (mutant 34). One hyperinvasive/adhesive mutant and four pst mutants that were pleiotropic and slightly attenuated in fish were also isolated.
-
-
-
Differential expression of mycobacterial proteins following phagocytosis by macrophages
More LessMycobacterium tuberculosis resides within the macrophages of the host, but the molecular and cellular mechanisms of survival are poorly understood. Recent evidence suggests that the attenuated vaccine strain Mycobacterium bovis BCG is both a deletion and regulatory mutant, yet retains both its immunoprotective and intra-macrophage survival potential. In an attempt to define M. bovis BCG genes expressed during interaction with macrophages, the patterns of protein synthesis were examined by both one- and two-dimensional gel electrophoresis of BCG while inside the human leukaemic macrophage cell line THP-1. This study demonstrated that BCG expresses proteins while resident inside macrophages that are not expressed during in vitro growth in culture media or under conditions of heat shock. Western blotting analysis revealed that some of the differentially expressed proteins are specifically recognized by human M. tuberculosis-infected sera. Proteome analysis by two-dimensional electrophoresis and MS identified six abundant proteins that showed increased expression inside macrophages: 16 kDa α-crystallin (HspX), GroEL-1 and GroEL-2, a 31·7 kDa hypothetical protein (Rv2623), InhA and elongation factor Tu (Tuf). Identification of proteins by such a strategy will help elucidate the molecular basis of the attenuation and the vaccine potential of BCG, and may provide antigens that distinguish infection with M. tuberculosis from vaccination with BCG.
-
- Physiology And Growth
-
-
-
Siderophore uptake and use by the yeast Saccharomyces cerevisiae
More LessThe non-reductive uptake of several siderophores (ferrioxamine B, ferrichrome, triacetylfusarinine C and ferricrocin) by various strains of Saccharomyces cerevisiae was studied. Several aspects of siderophore transport were examined, including specificity of transport, regulation of transport and intracellular localization of the ferri-siderophores. Ferrioxamine B was taken up preferentially via the products of the SIT1 gene and triacetylfusarinine C by the TAF1 gene product, but the specificity was not absolute. Ferrichrome and ferricrocin uptake was not dependent on a single major facilitator superfamily (MFS) gene product. The apparent specificity of transport was strongly dependent on the genetic background of the cells. Non-reductive uptake of siderophores was induced under more stringent conditions (of iron deprivation) than was the reductive uptake of ferric citrate. Regulation of transport depended on the transcriptional factors Aft1 and Tup1/Ssn6. Cells disrupted for the TUP1 or SSN6 genes were constitutively derepressed for the uptake of ferrichrome, ferricrocin or ferrioxamine B, but not for the uptake of triacetylfusarinine C. Cells bearing the AFT1 up mutation accumulated large amounts of ferric siderophores. Intracellular decomplexation of the siderophores occurred when transcription of the AFT1 up gene was repressed. Ferrioxamine B and ferrichrome seemed to accumulate in an endosomal compartment, as shown by biochemical studies and by confocal microscopy study of cells loaded with a fluorescent derivative of ferrichrome. Endocytosis was, however, not involved in the non-reductive uptake of siderophores.
-
-
-
-
Pyruvate kinase (Pyk1) levels influence both the rate and direction of carbon flux in yeast under fermentative conditions
More LessYeast phosphofructo-1-kinase (Pf1k) and pyruvate kinase (Pyk1) are allosterically regulated enzymes that catalyse essentially irreversible reactions in glycolysis. Both the synthesis and activity of these enzymes are tightly regulated. To separate experimentally the control of Pf1k and Pyk1 synthesis from their allosteric regulation, a congenic set of PFK1, PFK2 and PYK1 mutants was constructed in which these wild-type coding regions were driven by alternative promoters. Mutants carrying PGK1 promoter fusions displayed normal rates of growth, glucose consumption and ethanol production, indicating that the relatively tight regulation of Pyk1 and Pf1k synthesis is not essential for glycolytic control under fermentative growth conditions. Mutants carrying fusions to an enhancer-less version of the PGK1 promoter (PGK1 Δ767) expressed Pyk1 and Pf1k at about 2·5-fold lower levels than normal. Physiological and metabolic analysis of the PFK1 PFK2 double mutant indicated that decreased Pf1k had no significant effect on growth, apparently due to compensatory increases in its positive effector, fructose 2,6-bisphosphate. In contrast, growth rate and glycolytic flux were reduced in the PGK1 Δ767 –PYK1 mutant, which had decreased Pyk1 levels. Unexpectedly, the reduced Pyk1 levels caused the flow of carbon to the TCA cycle to increase, even under fermentative growth conditions. Therefore, Pyk1 exerts a significant level of control over both the rate and direction of carbon flux in yeast.
-
-
-
Genetic manipulation of 6-phosphofructo-1-kinase and fructose 2,6-bisphosphate levels affects the extent to which benzoic acid inhibits the growth of Saccharomyces cerevisiae
More LessThe mechanisms by which the weak acid preservative benzoic acid inhibits the growth of Saccharomyces cerevisiae have been investigated. A reduction in the pyruvate kinase level, which decreases glycolytic flux, did not increase the sensitivity of yeast to benzoic acid. However, a decrease in 6-phosphofructo-1-kinase (PF1K), which does not affect glycolytic flux, did increase sensitivity to benzoic acid. Also, resistance was increased by elevating PF1K levels. Hence, resistance to benzoic acid was not dependent upon optimum glycolytic flux, but upon an adequate PF1K activity. Benzoic acid was shown to depress fructose 2,6-bisphosphate levels in YKC14, a mutant with low PF1K levels. This effect was partially suppressed by overexpressing constitutively active 6-phosphofructo-2-kinase (Pfk26Asp644) or by inactivating fructose-2,6-bisphosphatase (in a Δfbp26 mutant). The inactivation of PF2K (in a Δpfk26 Δpfk27 mutant) increased benzoic acid sensitivity. Therefore, the antimicrobial effects of benzoic acid can be relieved, at least in part, by the genetic manipulation of PF1K or fructose 2,6-bisphosphate levels.
-
-
-
A purF mutant of Mycobacterium smegmatis has impaired survival during oxygen-starved stationary phase
More LessThe GenBank accession number for the sequence reported in this paper is AJ278609.
In this study it was demonstrated that a range of transposon mutants of Mycobacterium smegmatis, previously described as having impaired survival in carbon-starved stationary phase, were not markedly affected in O2-starved stationary-phase survival. One exception was 329B, a purine auxotroph, which showed a precipitous reduction in viability from
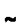 108 to
108 to 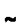 103 c.f.u. ml−1 during the first 5–10 d in O2-starved stationary phase. This was followed by an equally rapid recovery in culturability to a level within 10–100-fold of wild-type levels by 10–20 d into stationary phase. Transduction of the mutation into a clean genetic background demonstrated that the phenotype was due to the transposon insertion, which was shown to be in the purF gene. purF encodes phosphoribosylpyrophosphate amidotransferase, which catalyses the first committed step in purine biosynthesis. The M. smegmatis purF gene, which encodes a protein with a very high degree of similarity to the PurF homologues of Mycobacterium tuberculosis and Mycobacterium leprae, was cloned and shown to substantially complement the O2-starvation phenotype. The recovery in culturabilty of the purF mutant in O2-starved stationary phase did not involve movement of the transposon. In addition, when cells that had recovered culturability were retested, their survival kinetics in stationary phase were identical to the original culture, indicating that their recovery was not explained by the accumulation of suppressor mutations. It is concluded that the survival curve in O2-starved stationary phase for the purF mutant represents its true phenotype and is not a result of subsequent genetic changes in the culture. It is argued that the purF cells lose culturability for a finite period of time in stationary phase. Whether this is due to a fraction of the population dying and then regrowing using a previously undiscovered fermentation pathway, or becoming transiently dormant, or entering an active nonculturable state and subsequently undergoing resuscitation cannot be distinguished at this stage.
103 c.f.u. ml−1 during the first 5–10 d in O2-starved stationary phase. This was followed by an equally rapid recovery in culturability to a level within 10–100-fold of wild-type levels by 10–20 d into stationary phase. Transduction of the mutation into a clean genetic background demonstrated that the phenotype was due to the transposon insertion, which was shown to be in the purF gene. purF encodes phosphoribosylpyrophosphate amidotransferase, which catalyses the first committed step in purine biosynthesis. The M. smegmatis purF gene, which encodes a protein with a very high degree of similarity to the PurF homologues of Mycobacterium tuberculosis and Mycobacterium leprae, was cloned and shown to substantially complement the O2-starvation phenotype. The recovery in culturabilty of the purF mutant in O2-starved stationary phase did not involve movement of the transposon. In addition, when cells that had recovered culturability were retested, their survival kinetics in stationary phase were identical to the original culture, indicating that their recovery was not explained by the accumulation of suppressor mutations. It is concluded that the survival curve in O2-starved stationary phase for the purF mutant represents its true phenotype and is not a result of subsequent genetic changes in the culture. It is argued that the purF cells lose culturability for a finite period of time in stationary phase. Whether this is due to a fraction of the population dying and then regrowing using a previously undiscovered fermentation pathway, or becoming transiently dormant, or entering an active nonculturable state and subsequently undergoing resuscitation cannot be distinguished at this stage.
-
-
-
Alginate formation in Azotobacter vinelandii UWD during stationary phase and the turnover of poly-β-hydroxybutyrate
More LessAzotobacter vinelandii UWD is a mutant of strain UW that is defective in the respiratory oxidation of NADH. This mutation causes an overproduction of polyhydroxyalkanoates (PHAs), as polyester synthesis is used as an alternative electron sink. Since PHAs have potential for use as natural, biodegradable plastics, studies of physiology related to their production are of interest. Alginate production by this strain is limited to <11 μg (mg cell protein)−1, which permits high efficiency conversion of carbon source into PHA. However, ≤400 μg (mg cell protein)−1 was formed when UWD cells were oxygen-limited and in the stationary phase of growth. Alginate formation was fuelled by PHA turnover, which was coincident with the synthesis of alkyl resorcinols, under conditions of exogenous glucose limitation. However, alginate production was a phenotypic and reversible change. Alginate production was stopped by interruption of algD with Tn5lacZ. LacZ activity in UWD was shown to increase in stationary phase, while LacZ activity in a similarly constructed mutant of strain UW did not. Transcription of algD in strain UWD started from a previously identified RpoD promoter and not from the AlgU (RpoE) promoter. This is because strain UWD has a natural insertion element in algU. Differences between strain UW and UWD may reside in the defective respiratory oxidation of NADH, where the NADH surplus in strain UWD may act as a signal of stationary phase. Indeed, a backcross of UW DNA into UWD generated NADH-oxidase-proficient cells that failed to form alginate in stationary phase. Evidence is also presented to show that the RpoD promoter may be recognized by the stationary phase sigma factor (RpoS), which may mediate alginate production in strain UWD.
-
-
-
Catalase has a novel protective role against electrophile killing of Xanthomonas
More LessThe ability of Xanthomonas campestris pv. phaseoli to protect itself against lethal concentrations of man-made (N-ethylmaleimide, NEM) and endogenously produced (methylglyoxal, MG) electrophiles was investigated. Pretreatment of X. c. pv. phaseoli with a low concentration of NEM induced protection against lethal concentrations of NEM and MG. MG pretreatment weakly induced protection against NEM but not against MG itself. NEM-induced protection against electrophile killing required new protein synthesis and was abolished by the addition of a protein synthesis inhibitor. By contrast, MG-induced protection against NEM killing was independent of de novo protein synthesis. X. c. pv. phaseoli harbouring an expression vector carrying a catalase gene was over 100-fold more resistant to MG and NEM killing. High expression levels of genes for other peroxide-protective enzymes, such as those for alkyl hydroperoxide reductase (ahpC and ahpF) and ohr, failed to protect against electrophile killing. Thus, catalase appears to have a novel protective role(s) against electrophile toxicity. This finding suggests that in X. c. pv. phaseoli NEM and MG toxicity might involve accumulation and/or increased production of H2O2. This idea was supported by the observation that addition of 10 mM sodium pyruvate, a compound that can react chemically with peroxide or hydroxyl radical scavengers (DMSO and glycerol), was found to protect Xanthomonas from electrophile killing. The protective role of catalase and the role of H2O2 in electrophile toxicity are novel observations and could be generally important in other bacteria. In addition, unlike other bacteria, Xanthomonas in stationary phase was more susceptible to electrophile killing compared to cells in exponential phase.
-
- Plant-Microbe Interactions
-
-
-
In planta expression of a protein encoded by the extrachromosomal DNA of a phytoplasma and related to geminivirus replication proteins
The GenBank accession number for EcOYW1 is AB010426.
More LessA new extrachromosomal DNA, EcOYW1, was cloned from the onion yellows phytoplasma (OY-W). Southern blot and PCR analysis showed that EcOYW1 is not present in the OY-M, a mild symptom line derived from OY-W. We determined the complete nucleotide sequence of EcOYW1; it is a circular dsDNA of 7·0 kbp in length, which contains seven ORFs. ORF1 encoded a homologue of the geminivirus Rep protein. Western immunoblot analysis revealed that this Rep homologue is expressed in OY-W infected plants, suggesting that EcOYW1 replicates via a geminivirus-like rolling-circle replication mechanism. EcOYW1 is the first phytoplasmal extrachromosomal DNA shown to express encoded genes.
-
-
Volumes and issues
-
Volume 170 (2024)
-
Volume 169 (2023)
-
Volume 168 (2022)
-
Volume 167 (2021)
-
Volume 166 (2020)
-
Volume 165 (2019)
-
Volume 164 (2018)
-
Volume 163 (2017)
-
Volume 162 (2016)
-
Volume 161 (2015)
-
Volume 160 (2014)
-
Volume 159 (2013)
-
Volume 158 (2012)
-
Volume 157 (2011)
-
Volume 156 (2010)
-
Volume 155 (2009)
-
Volume 154 (2008)
-
Volume 153 (2007)
-
Volume 152 (2006)
-
Volume 151 (2005)
-
Volume 150 (2004)
-
Volume 149 (2003)
-
Volume 148 (2002)
-
Volume 147 (2001)
-
Volume 146 (2000)
-
Volume 145 (1999)
-
Volume 144 (1998)
-
Volume 143 (1997)
-
Volume 142 (1996)
-
Volume 141 (1995)
-
Volume 140 (1994)
-
Volume 139 (1993)
-
Volume 138 (1992)
-
Volume 137 (1991)
-
Volume 136 (1990)
-
Volume 135 (1989)
-
Volume 134 (1988)
-
Volume 133 (1987)
-
Volume 132 (1986)
-
Volume 131 (1985)
-
Volume 130 (1984)
-
Volume 129 (1983)
-
Volume 128 (1982)
-
Volume 127 (1981)
-
Volume 126 (1981)
-
Volume 125 (1981)
-
Volume 124 (1981)
-
Volume 123 (1981)
-
Volume 122 (1981)
-
Volume 121 (1980)
-
Volume 120 (1980)
-
Volume 119 (1980)
-
Volume 118 (1980)
-
Volume 117 (1980)
-
Volume 116 (1980)
-
Volume 115 (1979)
-
Volume 114 (1979)
-
Volume 113 (1979)
-
Volume 112 (1979)
-
Volume 111 (1979)
-
Volume 110 (1979)
-
Volume 109 (1978)
-
Volume 108 (1978)
-
Volume 107 (1978)
-
Volume 106 (1978)
-
Volume 105 (1978)
-
Volume 104 (1978)
-
Volume 103 (1977)
-
Volume 102 (1977)
-
Volume 101 (1977)
-
Volume 100 (1977)
-
Volume 99 (1977)
-
Volume 98 (1977)
-
Volume 97 (1976)
-
Volume 96 (1976)
-
Volume 95 (1976)
-
Volume 94 (1976)
-
Volume 93 (1976)
-
Volume 92 (1976)
-
Volume 91 (1975)
-
Volume 90 (1975)
-
Volume 89 (1975)
-
Volume 88 (1975)
-
Volume 87 (1975)
-
Volume 86 (1975)
-
Volume 85 (1974)
-
Volume 84 (1974)
-
Volume 83 (1974)
-
Volume 82 (1974)
-
Volume 81 (1974)
-
Volume 80 (1974)
-
Volume 79 (1973)
-
Volume 78 (1973)
-
Volume 77 (1973)
-
Volume 76 (1973)
-
Volume 75 (1973)
-
Volume 74 (1973)
-
Volume 73 (1972)
-
Volume 72 (1972)
-
Volume 71 (1972)
-
Volume 70 (1972)
-
Volume 69 (1971)
-
Volume 68 (1971)
-
Volume 67 (1971)
-
Volume 66 (1971)
-
Volume 65 (1971)
-
Volume 64 (1970)
-
Volume 63 (1970)
-
Volume 62 (1970)
-
Volume 61 (1970)
-
Volume 60 (1970)
-
Volume 59 (1969)
-
Volume 58 (1969)
-
Volume 57 (1969)
-
Volume 56 (1969)
-
Volume 55 (1969)
-
Volume 54 (1968)
-
Volume 53 (1968)
-
Volume 52 (1968)
-
Volume 51 (1968)
-
Volume 50 (1968)
-
Volume 49 (1967)
-
Volume 48 (1967)
-
Volume 47 (1967)
-
Volume 46 (1967)
-
Volume 45 (1966)
-
Volume 44 (1966)
-
Volume 43 (1966)
-
Volume 42 (1966)
-
Volume 41 (1965)
-
Volume 40 (1965)
-
Volume 39 (1965)
-
Volume 38 (1965)
-
Volume 37 (1964)
-
Volume 36 (1964)
-
Volume 35 (1964)
-
Volume 34 (1964)
-
Volume 33 (1963)
-
Volume 32 (1963)
-
Volume 31 (1963)
-
Volume 30 (1963)
-
Volume 29 (1962)
-
Volume 28 (1962)
-
Volume 27 (1962)
-
Volume 26 (1961)
-
Volume 25 (1961)
-
Volume 24 (1961)
-
Volume 23 (1960)
-
Volume 22 (1960)
-
Volume 21 (1959)
-
Volume 20 (1959)
-
Volume 19 (1958)
-
Volume 18 (1958)
-
Volume 17 (1957)
-
Volume 16 (1957)
-
Volume 15 (1956)
-
Volume 14 (1956)
-
Volume 13 (1955)
-
Volume 12 (1955)
-
Volume 11 (1954)
-
Volume 10 (1954)
-
Volume 9 (1953)
-
Volume 8 (1953)
-
Volume 7 (1952)
-
Volume 6 (1952)
-
Volume 5 (1951)
-
Volume 4 (1950)
-
Volume 3 (1949)
-
Volume 2 (1948)
-
Volume 1 (1947)
Most Read This Month


