
Full text loading...
A novel, strictly anaerobic, thermophilic, sulfate-reducing bacterium, designated strain AT1325T, was isolated from a deep-sea hydrothermal vent at the Rainbow site on the Mid-Atlantic Ridge. This strain was subjected to a polyphasic taxonomic analysis. Cells were Gram-negative motile rods (approximately 2.4×0.6 μm) with a single polar flagellum. Strain AT1325T grew at 55–75 °C (optimum, 65–70 °C), at pH 5.5–8.0 (optimum, 6.5–7.5) and in the presence of 1.5–4.5 % (w/v) NaCl (optimum, 2.5 %). Cells grew chemolithoautotrophically with H2 as an energy source and 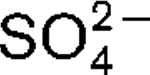 as an electron acceptor. Alternatively, the novel isolate was able to use methylamine, peptone or yeast extract as carbon sources. The dominant fatty acids (>5 % of the total) were C16 : 0, C18 : 1
ω7c, C18 : 0 and C19 : 0 cyclo ω8c. The G+C content of the genomic DNA of strain AT1325T was 45.6 mol%. Phylogenetic analyses based on 16S rRNA gene sequences placed strain AT1325T within the family Thermodesulfobacteriaceae, in the bacterial domain. Comparative 16S rRNA gene sequence analysis indicated that strain AT1325T belonged to the genus Thermodesulfatator, sharing 97.8 % similarity with the type strain of Thermodesulfatator indicus, the unique representative species of this genus. On the basis of the data presented, it is suggested that strain AT1325T represents a novel species of the genus Thermodesulfatator, for which the name Thermodesulfatator atlanticus sp. nov. is proposed. The type strain is AT1325T (=DSM 21156T=JCM 15391T).
as an electron acceptor. Alternatively, the novel isolate was able to use methylamine, peptone or yeast extract as carbon sources. The dominant fatty acids (>5 % of the total) were C16 : 0, C18 : 1
ω7c, C18 : 0 and C19 : 0 cyclo ω8c. The G+C content of the genomic DNA of strain AT1325T was 45.6 mol%. Phylogenetic analyses based on 16S rRNA gene sequences placed strain AT1325T within the family Thermodesulfobacteriaceae, in the bacterial domain. Comparative 16S rRNA gene sequence analysis indicated that strain AT1325T belonged to the genus Thermodesulfatator, sharing 97.8 % similarity with the type strain of Thermodesulfatator indicus, the unique representative species of this genus. On the basis of the data presented, it is suggested that strain AT1325T represents a novel species of the genus Thermodesulfatator, for which the name Thermodesulfatator atlanticus sp. nov. is proposed. The type strain is AT1325T (=DSM 21156T=JCM 15391T).

Article metrics loading...

Full text loading...
References


Data & Media loading...
Supplements
